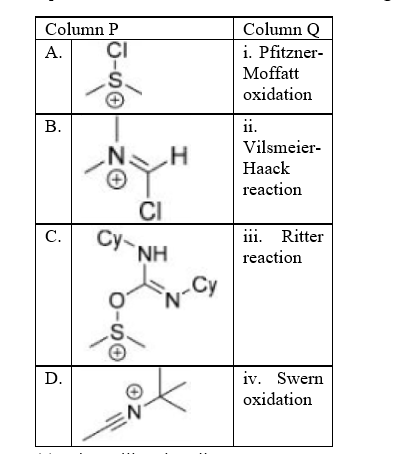
Correct option is D
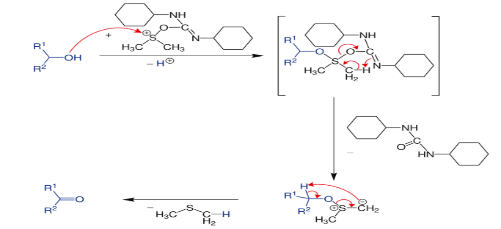
A. The Pfitzner–Moffatt oxidation, is a chemical reaction for the oxidation of primary and secondary alcohols to aldehydes and ketones, respectively. The oxidant is a combination of dimethyl sulfoxide (DMSO) and dicyclohexylcarbodiimide (DCC).
The reaction requires one equivalent each of the diimide, which is the dehydrating agent, and the sulfoxide, the oxidant:
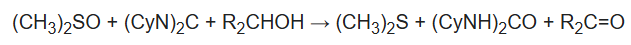
Typically, the sulfoxide and diimide are used in excess. The reaction cogenerates dimethyl sulfide and a urea. Dicyclohexylurea ((CyNH)2CO) can be difficult to remove from the product.
In terms of mechanism, the reaction is proposed to involve the intermediary of a sulfonium group, formed by a reaction between DMSO and the carbodiimide.
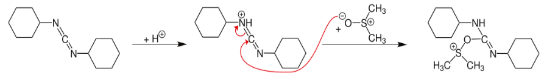
This species is highly reactive and is attacked by the alcohol. Rearrangement gives an alkoxysulfonium ylide which decomposes to give dimethyl sulfide and the carbonyl compound.
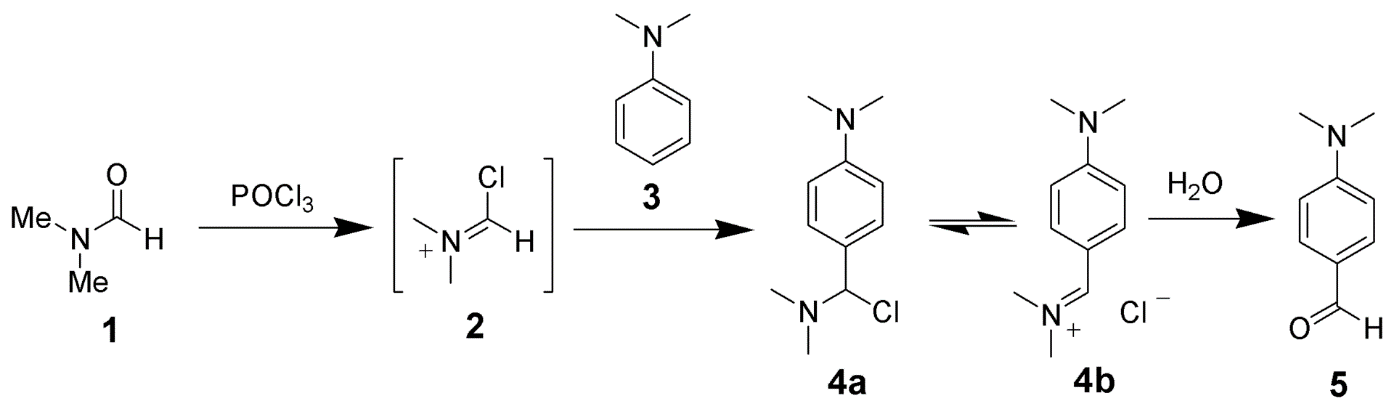
B. The Vilsmeier–Haack reaction (also called the Vilsmeier reaction) is the chemical reaction of a substituted formamide (1) with phosphorus oxychloride and an electron-rich arene (3) to produce an aryl aldehyde or ketone (5):
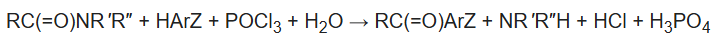
The reaction of a substituted amide with phosphorus oxychloride gives a substituted chloroiminium ion (2), also called the Vilsmeier reagent. The initial product is an iminium ion (4b), which is hydrolyzed to the corresponding ketone or aldehyde during workup.
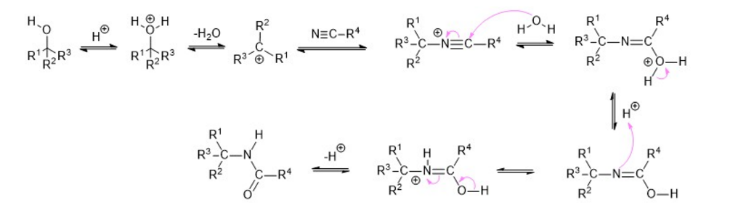
C. The Ritter reaction is a chemical reaction that transforms a nitrile into an N-alkyl amide using various electrophilic alkylating reagents. The original reaction formed the alkylating agent using an alkene in the presence of a strong acid.
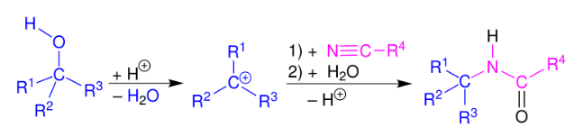
The Ritter reaction proceeds by the electrophilic addition of either a carbenium ion or covalent species to the nitrile. The resulting nitrilium ion is hydrolyzed to the desired amide.
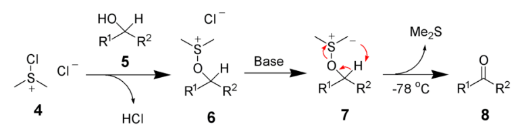
D. The Swern oxidation, is a chemical reaction whereby a primary or secondary alcohol (−OH) is oxidized to an aldehyde (−CH=O) or ketone (>C=O) using oxalyl chloride, dimethyl sulfoxide (DMSO) and an organic base, such as triethylamine.
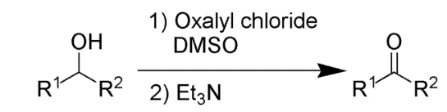
The first step of the Swern oxidation is the low-temperature reaction of DMSO, 1a, formally as resonance contributor 1b, with oxalyl chloride, 2. The first intermediate, 3, quickly decomposes giving off carbon dioxide and carbon monoxide and producing chloro(dimethyl)sulfonium chloride, 4.
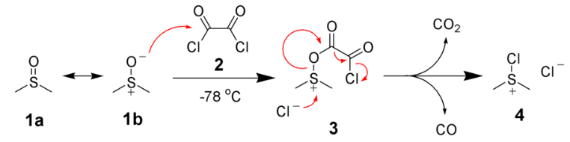
After addition of the alcohol 5, the chloro(dimethyl)sulfonium chloride 4 reacts with the alcohol to give the key alkoxysulfonium ion intermediate, 6. The addition of at least 2 equivalents of base -typically triethylamine- will deprotonate the alkoxysulfonium ion to give the sulfur ylide 7. In a five-membered ring transition state, the sulfur ylide 7 decomposes to give dimethyl sulfide and the desired carbonyl compound 8.