Correct option is D
The structures of many polynuclear organometallic species considers the total valence electron count, also called the Mingos cluster valence electron count.
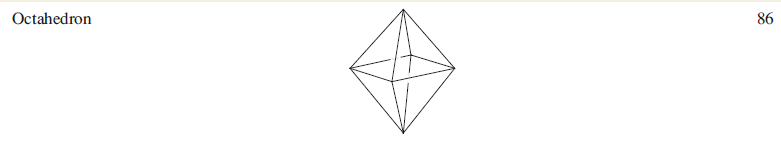
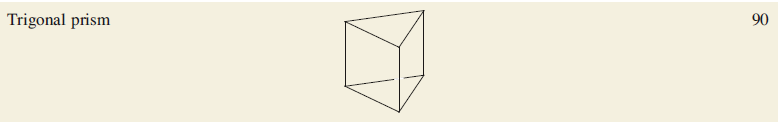
Each low oxidation state metal cluster cage possesses a characteristic number of valence electrons (ve). A and B are examples of a cage containing an interstitial atom which contributes all of its valence electrons to cluster bonding. An interstitial C atom contributes four electrons, a B atom three, an N or P atom five, and so on.
Each CO contributes two electrons.
Electronic configuration of Ru is ⦏Kr⦐4d⁷5s¹.
Electronic configuration of Os is [Xe] 4f145d66s2
A: Total valence electron count=8×6+2×17+3+1=86

B: Total valence electron count= 8×6+2×18+5+1=90
C: Total valence electron count= 8×4+2×16=64








