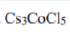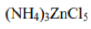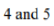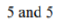Correct option is B
The coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it. The ion/molecule/atom surrounding the central ion/molecule/atom is called a ligand. This number is determined somewhat differently for molecules than for crystals.
For molecules and polyatomic ions the coordination number of an atom is determined by simply counting the other atoms to which it is bonded (by either single or multiple bonds).The common coordination numbers are 4, 6 and 8.
Cs3CoCl5 is of interest in inorganic chemistry as it provides a well defined example of a high-spin CoII complex with four monatomic ligands and only slightly distorted tetrahedral stereochemistry. The crystal structure of tri caesium cobalt pentachloride has been determined by X-ray diffraction at 10 K. It is made up of Cs+ cations, and [CoCl4]2-and Cl- anions. The [CoCl4]2- ion consists of a regular tetrahedron distorted along one of its axes.
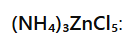
In this complex, the zinc ion (Zn²⁺) is tetrahedrally coordinated with four chloride ions.