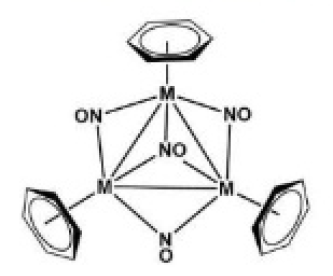
Correct option is D
Metal cluster compounds are a molecular ion or neutral compound composed of three or more metals and featuring significant metal-metal interactions.
Metal nitrosyl complexes are complexes that contain nitric oxide, NO, bonded to a transition metal. Many kinds of nitrosyl complexes are known, which vary both in structure and coligand.
The NO molecule can bind to a low oxidation state metal atom, and once coordinated NO is known as a nitrosyl ligand. Terminally bound NO can adopt two different bonding modes: linear or bent. In the linear mode, NO donates three electrons to the metal centre. Experimentally, a ‘linear’ MNO unit may have M–N–O bond angles in the range 165–180°, and in the IR spectrum, the vibrational wavenumber for the νNO mode lies in the approximate range 1650–1900 cm-1. In the bent mode, NO donates one electron to the metal centre. Bent nitrosyl ligands are characterized by having M–N–O bond angles in the range 120–140°, and in the IR spectrum, the νNO absorption typically lies between 1525 and 1690cm-1 .
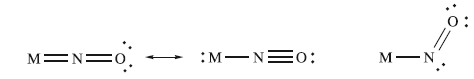
Nitrosyl ligands may also adopt bridging modes, acting as a 3-electron donor.
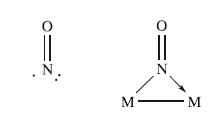
Benzene is an excellent ligand in the organometallic chemistry of low-valent metals. Benzene is a six-electron donor.
In the given compound, three benzene ligands donate eighteen electrons, four NO ligands donate twelve electrons and three Cr ([Ar] 3d⁵4s¹) atoms donate eighteen electrons. Thus, total electron count excluding the Cr-Cr bonds is 48.
Additional Knowledge
Option a: Three vanadium ([Ar] 3d³4s²) atoms donate fifteen electrons.
Option b: Three iron ([Ar] 3d⁶4s²) atoms donate 24 electrons.
Option c: Three manganese ([Ar] 4s²3d⁵) atoms donate 21 electrons.