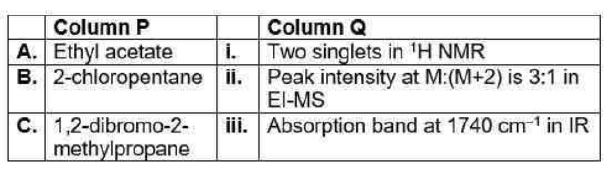
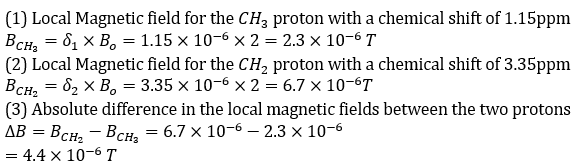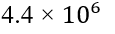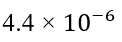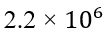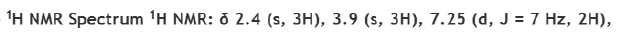Correct option is C
The local magnetic field experienced by a proton in NMR depends on its chemical shift and the external magnetic field applied. The relationship between the local magnetic field (Blocal) and the chemical shift (δ) is:
Formula: Blocal = δ x Bo
Chemical Shift: The chemical shift is measured in parts per million (ppm), and it reflects the difference in the local magnetic field experienced by different protons in the molecule.
External Magnetic Field: The external magnetic field (Bo) is given in Tesla (T). In this case, Bo = 2T.
Local Magnetic Field: The local magnetic field experienced by each proton can be found by multiplying the chemical shift by the external magnetic field.
Absolute Difference: The absolute difference in local magnetic fields between two protons can be calculated by subtracting the local magnetic fields calculated for each proton.