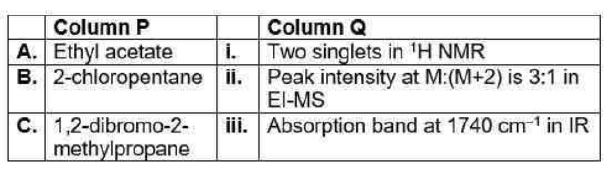
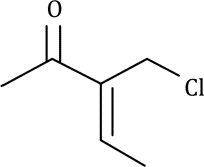
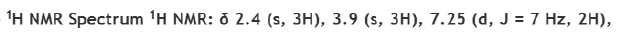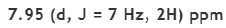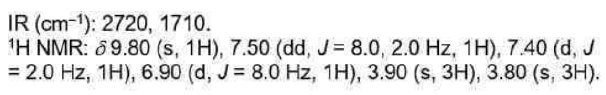Correct option is C
Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of infrared radiation with matter by absorption, emission, or reflection. It is used to study and identify chemical substances or functional groups in solid, liquid, or gaseous forms. It can be used to characterize new materials or identify and verify known and unknown samples.
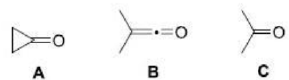
Ketones show a very strong band for the C=O group that appears in the range of 1720–1708 cm-1.
The s character in the C=O group increases as the ring size decreases.
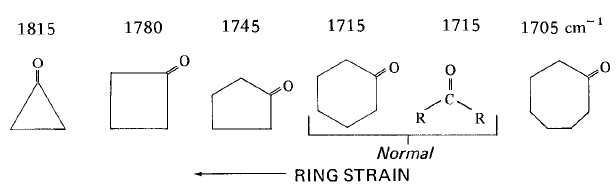
Ring strain is a type of instability that exists when bonds in a molecule form angles that are abnormal. Strain is most commonly discussed for small rings such as cyclopropanes and cyclobutanes, whose internal angles are substantially smaller than the idealized value of approximately 109°.

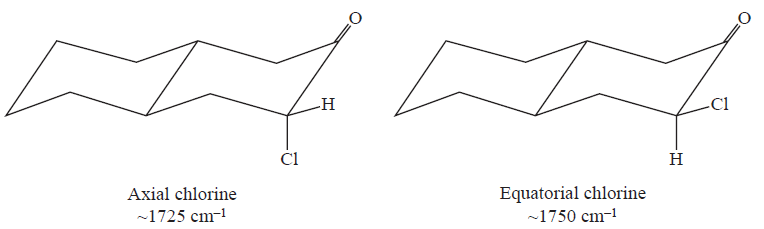
When the carbon next to the carbonyl is substituted with a chlorine (or other halogen) atom, the carbonyl band shifts to a higher frequency. The electron-withdrawing effect removes electrons from the carbon of the C=O bond. This removal is compensated for by a tightening of the pi bond (shortening), which increases the force constant and leads to an increase in the absorption frequency. This effect holds for all carbonyl compounds.
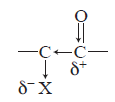
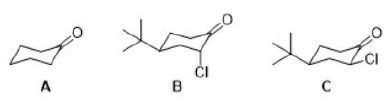
In ketones, two bands result from the substitution of an adjacent chlorine atom. One arises from the conformation in which the chlorine is rotated next to the carbonyl, and the other is due to the conformation in which the chlorine is away from the group. When the chlorine is next to the carbonyl, nonbonded electrons on the oxygen atom are repelled, resulting in a stronger bond and a higher absorption frequency. Information of this kind can be used to establish a structure in rigid ring systems, such as in the following examples: