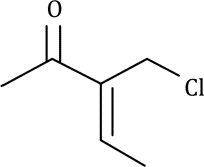
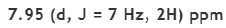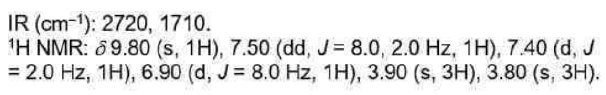Correct option is D
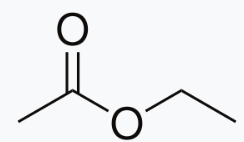
Ethyl acetate:
Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of infrared radiation with matter by absorption, emission, or reflection. It is used to study and identify chemical substances or functional groups in solid, liquid, or gaseous forms.
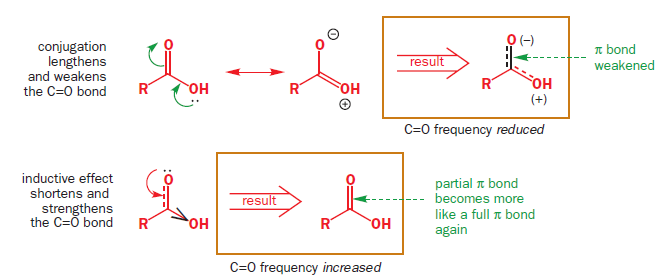
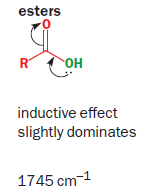
A much better measure is the difference in IR stretching frequency of the C=O group. Esters have a well balanced combination with the inductive effect slightly stronger (oxygen donates from a compatible 2p orbital but is very electronegative and so withdraws electrons strongly
as well).
2-chloropentane:
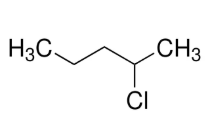
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a mass spectrum, a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used in many different fields and is applied to pure samples as well as complex mixtures. In a typical MS procedure, a sample, which may be solid, liquid, or gaseous, is ionized, for example by bombarding it with a beam of electrons. This may cause some of the sample's molecules to break up into positively charged fragments or simply become positively charged without fragmenting. These ions (fragments) are then separated according to their mass-to-charge ratio, for example by accelerating them and subjecting them to an electric or magnetic field: ions of the same mass-to-charge ratio will undergo the same amount of deflection. The ions are detected by a mechanism capable of detecting charged particles, such as an electron multiplier. Results are displayed as spectra of the signal intensity of detected ions as a function of the mass-to-charge ratio. The atoms or molecules in the sample can be identified by correlating known masses (e.g. an entire molecule) to the identified masses or through a characteristic fragmentation pattern.
Peaks caused by ions bearing those heavier isotopes also appear in mass spectra. The relative abundances of such isotopic peaks are proportional to the abundances of the isotopes in nature. Most often, the isotopes occur one or two mass units above the mass of the “normal” atom. Therefore, besides looking for the molecular ion (M+) peak, one would also attempt to locate M + 1 and M + 2 peaks.
Natural abundance: 35Cl=75% and 37Cl=25%
Relative natural abundance: 35Cl=(75/75)×100=100% and 37Cl=(25/75)×100=33%
Intensity ratio: M:M+2
35Cl:37Cl
100:33
(100/33):(33/33)=3:1
1,2-dibromo-2-methylpropane
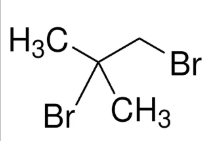
Nuclear magnetic resonance (NMR) spectroscopy is a resonance technique involving absorption of radiofrequency energy. The magnetic environment of a nucleus affects its resonance frequency and allows structural information to be deduced.
Structural assignment is often helped by the observation of the spin–spin coupling, which gives rise to multiplets in the spectrum due to interactions between nuclear spins. Spin–spin coupling arises when the orientation of the spin of a nearby nucleus affects the energy of another nucleus and causes small changes in the location of the latter’s resonance. A multiplet of 2I+1 lines is obtained when a spin-1/2 nucleus (or a set of symmetry-related spin-1/2 nuclei) is coupled to a nucleus of spin I. The coupling of the nuclear spins of different elements is called heteronuclear coupling. Homonuclear coupling between nuclei of the same element is detectable when the nuclei are in chemically inequivalent locations.
A singlet will be observed due to six protons (as they are in the same environment). Another singlet will be observed due to two protons.