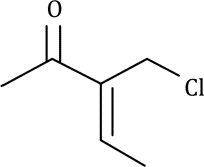
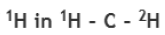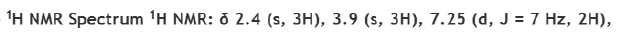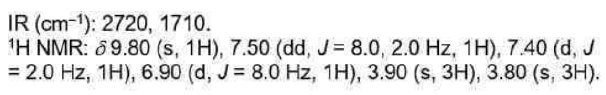Correct option is C
Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy or magnetic resonance spectroscopy (MRS), is a spectroscopic technique based on re-orientation of atomic nuclei with non-zero nuclear spins in an external magnetic field. This re-orientation occurs with absorption of electromagnetic radiation in the radio frequency region from roughly 4 to 900 MHz, which depends on the isotopic nature of the nucleus and increased proportionally to the strength of the external magnetic field. Notably, the resonance frequency of each NMR-active nucleus depends on its chemical environment. As a result, NMR spectra provide information about individual functional groups present in the sample, as well as about connections between nearby nuclei in the same molecule. As the NMR spectra are unique or highly characteristic to individual compounds and functional groups, NMR spectroscopy is one of the most important methods to identify molecular structures, particularly of organic compounds.
NMR-active nuclei, particularly those with a spin quantum number of 1/2, are of great significance in NMR spectroscopy. Examples include 1H, 13C, 15N, and 31P.
The energy difference ΔE between nuclear spin states is proportional to the magnetic field. ΔE is also sensitive to electronic environment of the nucleus, giving rise to what is known as the chemical shift, δ.
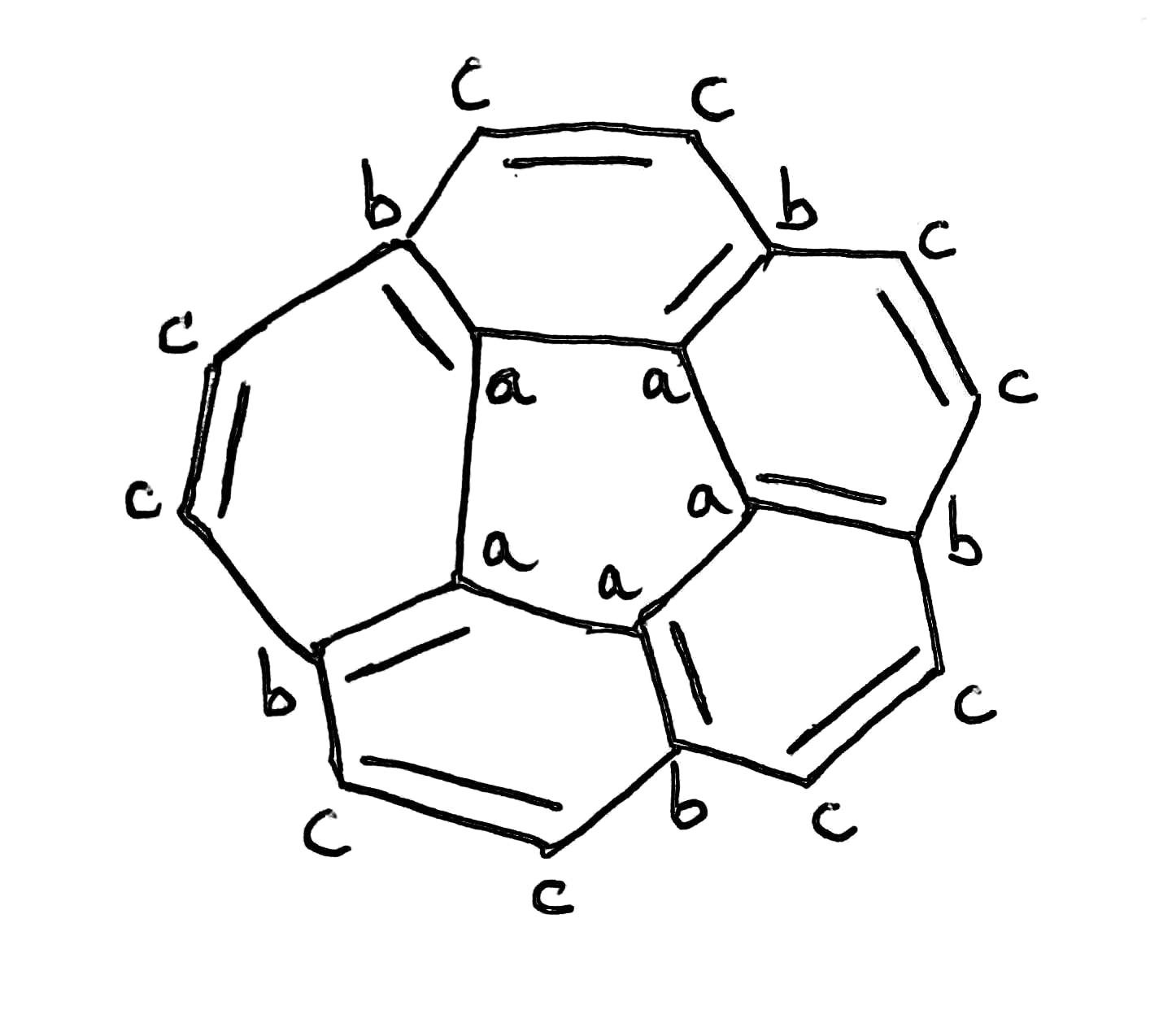
All of the carbon atoms found in chemically identical environments within a molecule are chemically equivalent, and they often exhibit the same chemical shift. Each such compound gives rise to a single absorption peak in its NMR spectrum. On the other hand, nearly every nonequivalent carbon atom in an organic molecule gives rise to a peak with a different chemical shift.
Due to similar chemical environment, carbon a are of same type.
Carbon b are of same type.
Carbon c are of same type.