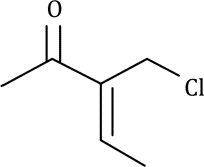
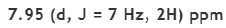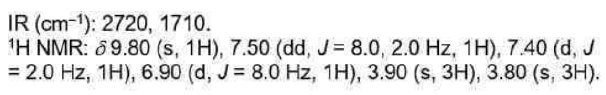Correct option is D
Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of infrared radiation with matter by absorption, emission, or reflection. It is used to study and identify chemical substances or functional groups in solid, liquid, or gaseous forms. It can be used to characterize new materials or identify and verify known and unknown samples. The method or technique of infrared spectroscopy is conducted with an instrument called an infrared spectrometer (or spectrophotometer) which produces an infrared spectrum. An IR spectrum can be visualized in a graph of infrared light absorbance (or transmittance) on the vertical axis vs. frequency, wavenumber or wavelength on the horizontal axis. Typical units of wavenumber used in IR spectra are reciprocal centimeters, with the symbol cm-1.
The carbonyl group is present in aldehydes, ketones, acids, esters, amides, acid chlorides, and anhydrides. This group absorbs strongly in the range from 1850 to 1650 cm-1 because of its large change in dipole moment.
C=O stretch appears in the range 1720-1708 cm-1 for normal aliphatic ketones.
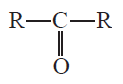
C=O stretch appears at 1815 cm-1 for cyclopropanone.
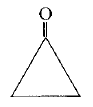
C=O stretch appears at 2140 cm-1 for ketene.
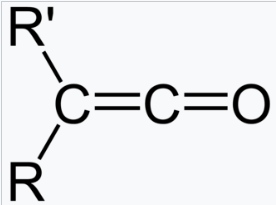
The carbonyl stretching frequency of a molecule increases with increase in the % s character in the C=O bond.
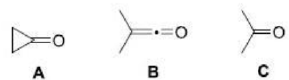
In compounds A, B, and C, the carbon atom of the carbonyl group (C=O) is sp2, sp, and sp2 hybridized respectively.
In sp hybridization, s and p each contribute 50 %. In sp2 hybridization, s contributes 33.33%, while p contributes 66.66%.
Compound B has the highest carbonyl stretching frequency as the % s character is the highest.
Cyclic ketones: Ring strain shifts the absorption values to a higher frequency. Ring strain is a type of instability that exists when bonds in a molecule form angles that are abnormal. Strain is most commonly discussed for small rings such as cyclopropanes and cyclobutanes, whose internal angles are substantially smaller than the idealized value of approximately 109°.