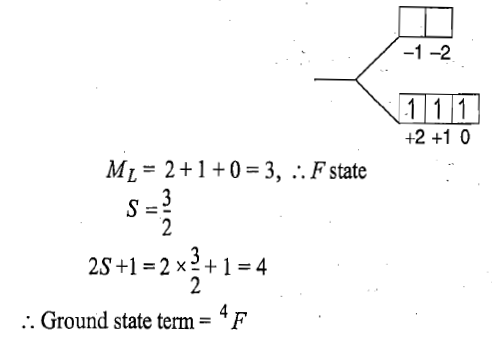
Correct option is B
In d3octahedral complexes the d-orbitals split into t2g (lower energy) and eg (higher energy) orbital. Therefore, in ground state the electronic configuration is
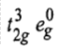
and there are two holes in eg orbitals.
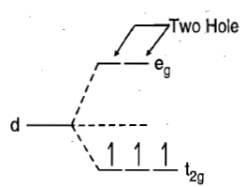
There is only one electronic arrangement corresponding to this electronic configuration
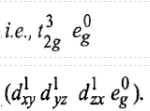
Therefore, this arrangement is electronically non-degenerate and can be represented as
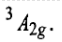
This case is similar to d8 octahedral complex. When an electron is excited to any one of the eg orbitals, the electronic configuration becomes
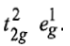
Now there are two holes, one in t2g and one in eg orbitals. There are two triply degenerate arrangements of holes corresponding to this electronic configuration. The lower energy state will arise when two holes occupy the orbitals as far apart as possible. Therefore, the two holes may be present either in
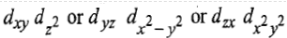
Thus, this state is triply degenerate and is represented as
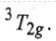

Therefore, there are three transitions of holes from ground states to three other excited states. Transition of holes is similar to electronic transition.

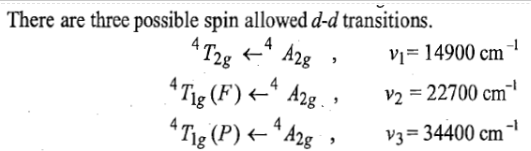
22700cm-1=440nm