Correct option is A
Proton nuclear magnetic resonance or 1H NMR is the application of nuclear magnetic resonance in NMR spectroscopy with respect to hydrogen nuclei within the molecules of a substance, in order to determine the structure of its molecules.
Proton NMR spectra are characterized by chemical shifts in the range of +14 to -4ppm and by spin-spin coupling between protons. The integration curve for each proton reflects the abundance of the individual protons.

This is because the five equivalent protons of the
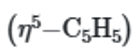
ring will show one singlet in 1H NMR spectra.
While, the
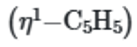
ring will also show one singlet in 1H NMR spectra, although the protons are not all equivalent.
Ring whizzing
The compound
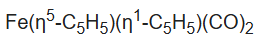
exhibits the phenomenon of "ring whizzing". At 30 °C, the 1H NMR spectrum shows only two peaks, one typical

and the other assigned
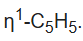
The singlet assigned to the
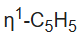
ligand splits at low temperatures owing to the slow hopping of the Fe center from carbon to carbon in the
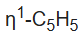
ligand. Two mechanisms have been proposed, with the consensus favoring the 1,2 shift pathway.
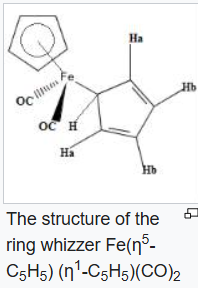
By the ring whizzer mechanism, the five ring positions of the monohapto ring of
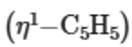
interchange via 1,2-metal shift so rapidly at room temperature that the NMR spectrometer can detect only the average signal for the ring.
At lower temperature, this process is slower and the different resonances for the protons of
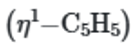
become apparent at lower temperatures, the peak at 4.5ppm
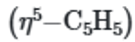
remains constant, but the other peak at 5.7ppm for
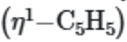
spreads and then splits into four peaks of intensity ratio (5:2:2:1).







