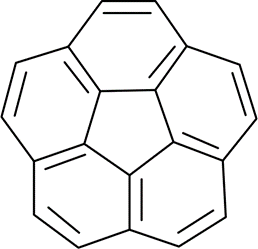
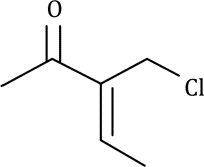
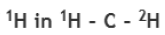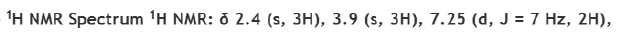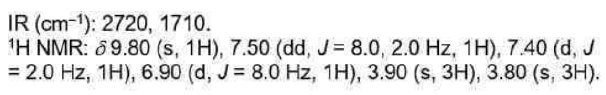Correct option is D
Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy, is a spectroscopic technique based on re-orientation of atomic nuclei with non-zero nuclear spins in an external magnetic field. This re-orientation occurs with absorption of electromagnetic radiation in the radio frequency region from roughly 4 to 900 MHz, which depends on the isotopic nature of the nucleus and increased proportionally to the strength of the external magnetic field.
A type of information to be found in the NMR spectrum is that derived from the spin–spin splitting phenomenon. For instance, in 1,1,2-trichloroethane there are two chemically distinct types of hydrogens:
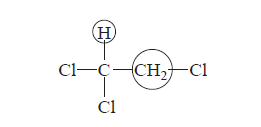
The high-resolution NMR spectrum of this compound has five peaks: a group of three peaks (called a triplet) at 5.77 ppm and a group of two peaks (called a doublet) at 3.95 ppm.The methine (CH) resonance (5.77 ppm) is said to be split into a triplet, and the methylene resonance (3.95 ppm) is split into a doublet. This phenomenon, called spin–spin splitting, can be explained empirically by the so-called n + 1 Rule. Each type of proton “senses” the number of equivalent protons (n) on the carbon atom(s) next to the one to which it is bonded, and its resonance peak is split into (n + 1) components.
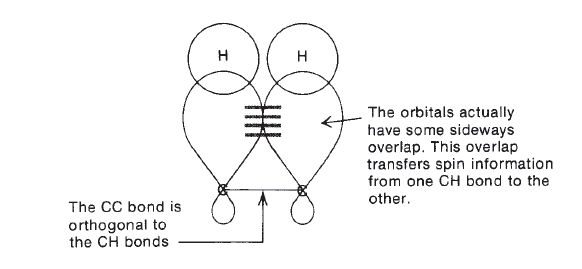
J-couplings are mediated through chemical bonds connecting two spins. It is an indirect interaction between two nuclear spins that arises from hyperfine interactions between the nuclei and local electrons. In NMR spectroscopy, J-coupling contains information about relative bond distances and angles. Most importantly, J-coupling provides information on the connectivity of chemical bonds. The J coupling (always reported in Hz) is field-independent (i.e. J is constant at different external magnetic field strength), and is mutual (i.e. JAX = JXA).
The
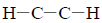
couplings are usually called vicinal couplings because the hydrogens are on neighboring carbon atoms (Latin vicinus = “neighbor”). Vicinal couplings are three-bond couplings and have a coupling constant designated as 3J.These couplings produce spin–spin splitting patterns that follow the n + 1 Rule in simple aliphatic hydrocarbon chains.
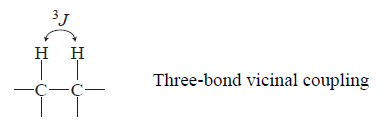
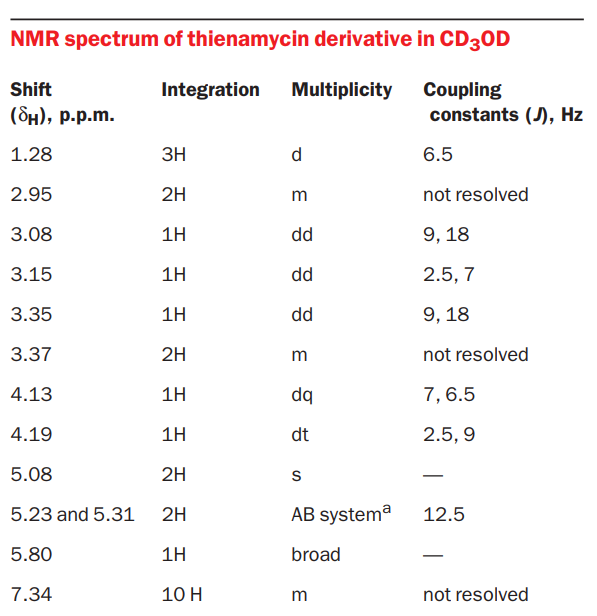
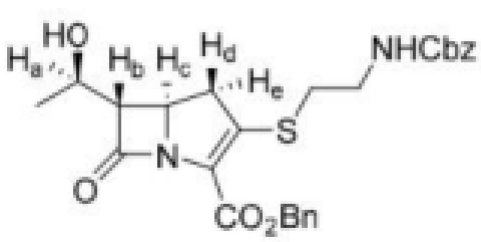
The NMR spectrum of a thienamycin derivative with protecting groups on the amine and carboxylic acids is shown below.
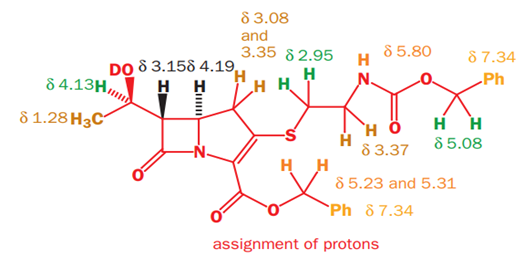
Proton Hb will couple with the adjacent protons Ha and Hc to give a doublet and a doublet respectively. Proton Ha will couple with adjacent protons of the methyl group and Hb to give a quartet and a doublet respectively. Proton Hc will couple with adjacent Hb and Hd (and He) to give a doublet and a triplet respectively. Hd and He will couple with each other to give a doublet. Hd and He will also couple with Hc to give a doublet.