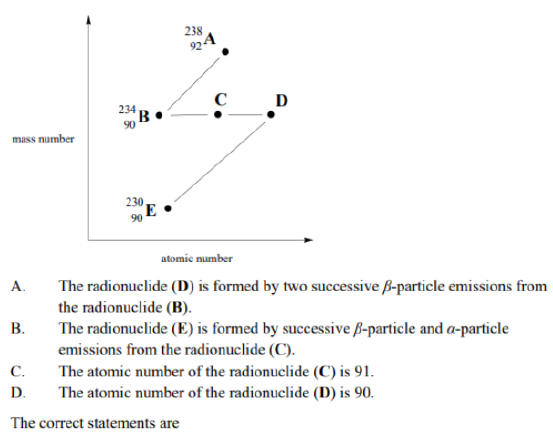
Correct option is D
Beta decay (β- decay) is a type of radioactive decay in which an atomic nucleus emits a beta particle (fast energetic electron or positron), transforming into an isobar of that nuclide.
The two types of beta decay are known as beta minus and beta plus. In beta minus (β-) decay, a neutron is converted to a proton, and the process creates an electron and an electron antineutrino; while in beta plus (β+) decay, a proton is converted to a neutron and the process creates a positron and an electron neutrino. β+ decay is also known as positron emission.
An example of electron emission (β- decay) is the decay of carbon-14 into nitrogen-14 with a half-life of about 5,730 years:
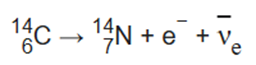
In this form of decay, the original element becomes a new chemical element in a process known as nuclear transmutation. This new element has an unchanged mass number A, but an atomic number Z that is increased by one.
Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or "decays" into a different atomic nucleus, with a mass number that is reduced by four and an atomic number that is reduced by two.
C has an atomic number of 91. (Statement C)
D has an atomic number of 92 (Statement A)
E has an atomic number of 90 (Statement B)