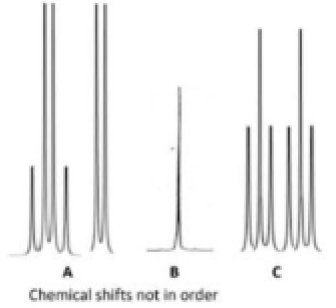
Correct option is A
Phosphorus-31 NMR spectroscopy is an analytical chemistry technique that uses nuclear magnetic resonance (NMR) to study chemical compounds that contain phosphorus.
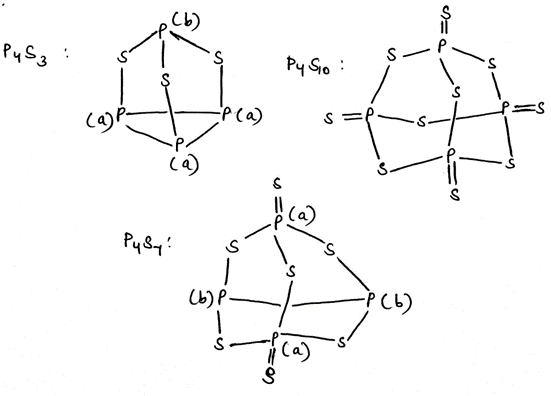
In
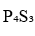
, phosphorus atoms of type a will give a doublet by coupling with phosphorus atom of type b. Similarly, phosphorus atom of type b will give a quartet due to three phosphorus atoms of type a.
In
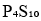
, all the phosphorus atoms are same and a singlet will be observed.
In
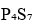
, two phosphorus atoms of type a will give a triplet by coupling with two phosphorus atoms of type b. Also, two phosphorus atoms of type b will give a triplet by coupling with two phosphorus atoms of type a.