Correct option is C
When CoCl₂ reacts with KNO₂ in an acidic medium, it forms a yellow precipitate, K₃[Co(NO₂)₆]. Upon reaction with NH₄SCN, a blue-colored compound (NH₄)₂[Co(SCN)₄] is formed, which is characteristic of cobalt thiocyanate complexes.
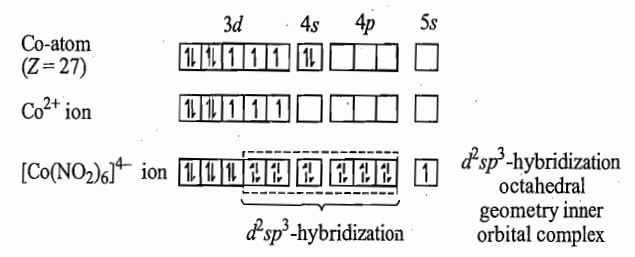
[Co(NO2)6]4- ion: In this complex ion, oxidation state of cobalt is +2 and its valence shell electronic configuration is 3d7 .The (NO2)- ligands are strong, they, therefore, cause pairing of metal 3d- electrons. Pauling suggested that two vacant 3d-orbitals are made available by promotion of an unpaired electron from a 3d-orbital to 5s-orbital.
The presence of an unpaired electron in 5s-orbital is supported by the fact that, 5s-orbital has very high energy and the electron present in it is loosely bound and can be removed easily. Experimentally it is also observed that [Co(NO2)6]4- is oxidized by air or H2O2 easily to give [Co (NO2)6]3- .This indicates that the complex [Co(NO2)6]4- is unstable in air. Thus, this complex should be prepared in inert atmosphere.