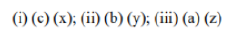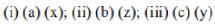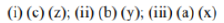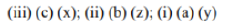Correct option is A
Electronic configuration of W is:

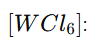
Let the oxidation state of W be x.
x-6=0,x=+6
Total d electrons=0
Each chloride ion donates one electron.
Total valence electrons=6+6(1)=12
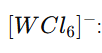
Let the oxidation state of W be x.
x-6=-1,x=+5
Total d electron=1
Each chloride ion donates one electron.
Total valence electrons=6+6(1)+1=13
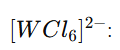
Let the oxidation state of W be x.
x-6=-2,x=+4
Total d electrons=2
Each chloride ion donates one electron.
Total valence electrons=6+6(1)+2=14