Correct option is D
Charge-transfer bands are a characteristic feature of the optical spectra of many compounds. These bands are typically more intense than d–d transitions. CT absorptions bands are intense and often lie in the ultraviolet or visible portion of the spectrum. For coordination complexes, charge-transfer bands often exhibit molar absorptivities, ε, of about 50000 L mol-1cm-1.By contrast ε values for d–d transitions are in the range of 20–200 L mol-1cm-1.CT transitions are spin-allowed and Laporte-allowed. The weaker d–d transitions are potentially spin-allowed but always Laporte-forbidden.
The electronic transitions between d orbitals that is t2g to eg in octahedral complexes or e to t2 in tetrahedral complexes are called d-d transitions. When d-d transitions occur in complexes with centre of symmetry such as regular octahedral complexes, the intensities of absorption bands are low. But when d-d transitions occur in complexes which lack centre of symmetry such as tetrahedral complexes, relatively strong absorption bands are observed.
According to the Laporte selection rule, the transitions that occur between states of same parity i.e. g↔g or u↔u are forbidden. For example, d-d transitions are forbidden because d-orbitals are gerade, i.e. these have centre of symmetry with respect to wave function.
In tetrahedral complexes there is no centre of symmetry and p-d mixing is more pronounced in tetrahedral complexes because t2 molecular orbitals are formed from atomic d (gerade) and p (ungerade) orbitals. Tetrahedral complexes, therefore, absorb more strongly than octahedral complexes. Therefore, the tetrahedral complexes give more intense colour than octahedral complexes.
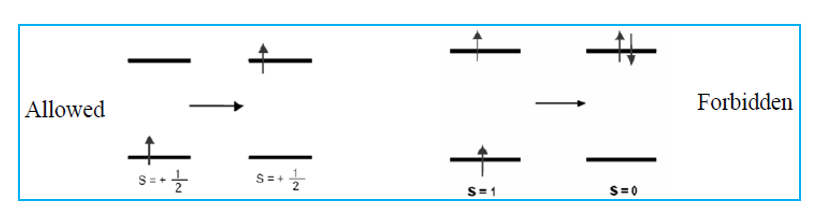
During electronic transition if spin of the electron is not changed, then the transition is spin allowed. This is because an electromagnetic radiation usually cannot change the relative orientation of an electron spin.
In [TiCl6]2-,the oxidation state of Ti is four and Cl- is a pi donor ligand. A pi-donor ligand is a kind of ligand endowed with filled non-bonding orbitals that overlap with metal-based orbitals. If the transfer occurs from the MO with ligand-like character to the metal-like one, the transition is called a ligand-to-metal charge-transfer (LMCT).
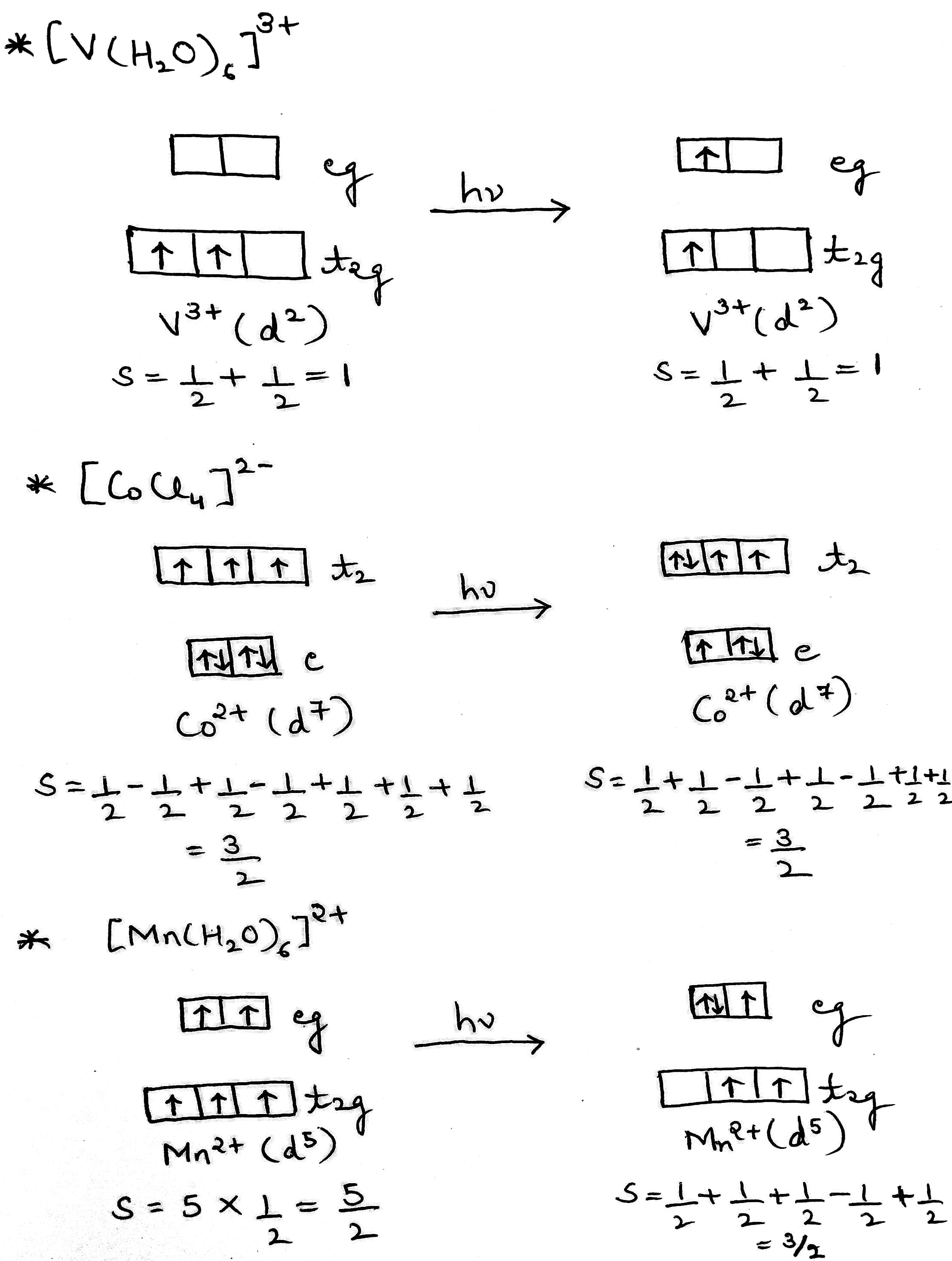
Complex b (V3+) is Laporte forbidden and spin allowed.
Complex c (Co2+) is Laporte partly allowed and spin allowed.
Complex d (Mn2+) is both Laporte and spin forbidden.