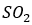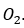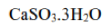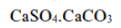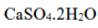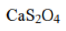Correct option is C
Current awareness of the effects of environmental pollution has been instrumental in the development of desulfurization processes. This includes desulfurization of fossil fuels and flue gases from a variety of sources.
The aim in a flue gas desulfurization process is to optimize the removal of SO2 from emissions into the atmosphere. One important method of desulfurization in commercial operation throughout the world is based upon the neutralization reactions between Ca(OH)2 or CaCO3 and sulfuric acid. General equations for the neutralization reactions are:
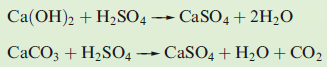
The reactions shown below remove >90% of the SO2 produced at a power station.
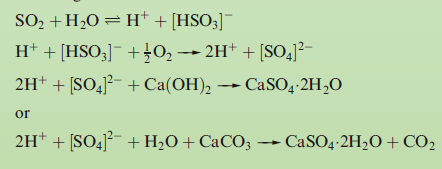
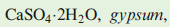
is non-toxic and is not a waste product. It has a number of commercial applications, for example in the production of plaster of Paris and cement.