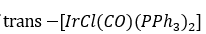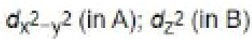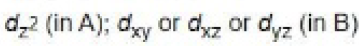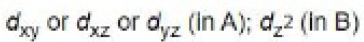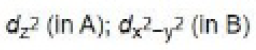Correct option is C
Oxidative addition reactions are very important in organometallic synthesis.
The oxidation number on the metal atom increased by 2 when the dihydrogen reacted to give a dihydride:
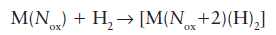
The increase in oxidation number of the metal by 2 arises because dihydrogen is treated as a neutral ligand, whereas the hydride ligands are treated as H- : thus the formation of two M-H bonds from a H2 molecule corresponds to a formal increase in the charge on the metal by 2. Whilst it might seem that this oxidation of the metal is just an anomaly thrown up by the method of counting electrons, two of the electrons on the metal atom have been used to backbond to the dihydrogen, and these two electrons are no longer available to the metal for further bonding. This type of reaction is quite general and is known as oxidative addition. A large number of molecules add oxidatively to a metal atom, including the alkyl and aryl halides, dihydrogen, and simple hydrocarbons. In general, the addition of any molecule X-Y to a metal atom, where both X and Y are more electronegative than the metal, can be classed as oxidative addition.
Oxidative addition reactions result in two more ligands bound to the metal with an increase in the total electron count at the metal of 2. Thus oxidative addition reactions normally require a coordinatively unsaturated metal centre, and are particularly common for 16-electron square-planar metal complexes.
The oxidative addition of hydrogen is a concerted reaction: dihydrogen coordinates to form a σ-bonded H2 ligand, and then backbonding from the metal results in cleavage of the H-H bond and the formation of cis dihydrides:
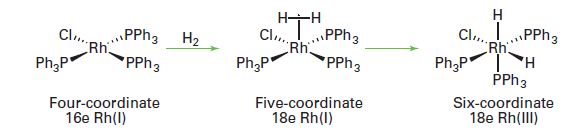
Other molecules, such as alkanes and aryl halides, are known to react in a concerted fashion, and in all these cases the two incoming ligands end up cis to each other.
Some oxidative addition reactions are not concerted and either go through radical intermediates or are best thought of as SN2 displacement reactions. In an SN2 oxidative addition reaction, a lone pair on the metal attacks the X-Y molecule displacing Y- , which subsequently bonds to the metal:
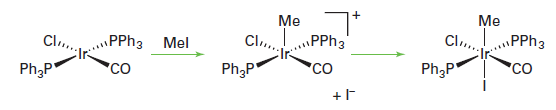
There are two stereochemical consequences of this reaction. First, the two incoming ligands need not end up cis to each other and, second, unlike the concerted reaction, any chirality at the X group is inverted. An SN2 -type oxidative addition is common for polar molecules such as alkyl halides.
Hybrid orbitals may be formed by mixing the characters of atomic orbitals that are close in energy. The character of a hybrid orbital depends on the atomic orbitals involved and their percentage contributions. Hybridization of
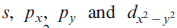
atomic orbitals gives four sp2d hybrid orbitals corresponding to a square planar arrangement.
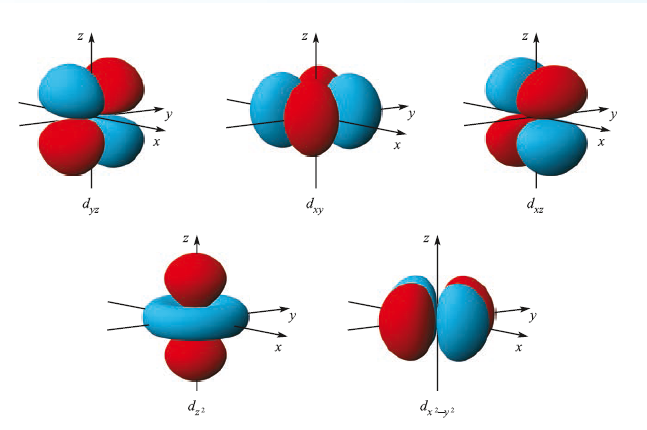
In the path B, the dz2 orbital is involved in the electron transfer from iridium to MeI as it is perpendicular to dx2 -y2 orbital.
In path A, dxy (or dxz or dyz) orbital is involved in the electron transfer from iridium to H2.