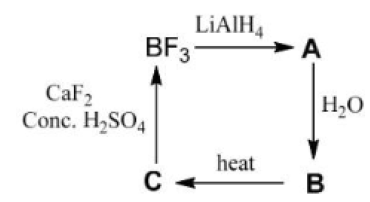
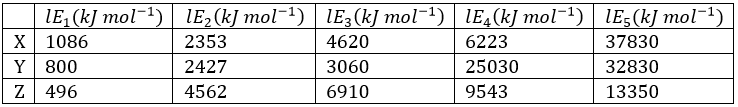
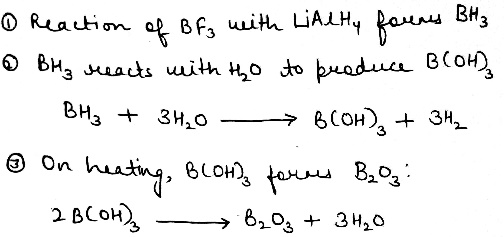Correct option is D
B₂O₃ is a stable oxide of boron widely used in glass and ceramics. BH₃ is a Lewis acid and exists in dimeric form as diborane (B₂H₆). B(OH)₃ is weakly acidic and has uses in buffer solutions and antiseptics.
Additional Knowledge:
1. LiBH₄ (Lithium borohydride) is a reducing agent but not a product here.
2. Amorphous boron and crystalline B₁₂ represent other allotropes but are unrelated in this context.