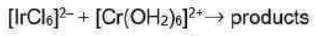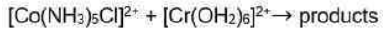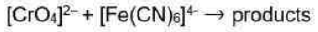Correct option is D
In inner sphere electron transfer reactions, the oxidant and reductant share a ligand in their coordination sphere to form a bridged complex, the electron is then transferred through the bridging ligand.
Salient Features of Inner Sphere Mechanism
(1) One complex (the reductant) is labile and the other (i.e., the oxidant) is inert.
(2) The inert complex possesses atleast one ligand capable of bridging two metal ions to form the bridged intermediate and this bridged intermediate is called a precursor complex.
(3) Often, but not always, the bridging ligand is also transferred from oxidant to reductant. The transfer or non-transfer of bridging ligand depends upon the relative stabilities of the product. The ligand transfer is a good indicator that electron transfer takes place by inner sphere mechanism. If there is no bridging ligand, then electron transfer does not take place by inner sphere mechanism.
(4) Either eg (σ*) or t2g (π*) orbitals of both the reactants participate in electron transfer by inner sphere mechanism. In general these orbitals may be HOMO of the reductant and LUMO of the oxidant. If both the reactants in an electron transfer reaction involve orbitals of same symmetry, no or a little activation energy is required and electron transfer will be fast. If both the orbitals are of different symmetries, greater activation energy encompassing both structural deformation and electron configuration change is required. Such reaction will be slower than those requiring no electron configuration change. Electron transfer by inner sphere mechanism is faster when electron transfer takes place between eg orbitals of oxidant and reductant.
(5) The rate of electron transfer increases if the bridging ligand possess unsaturation or extended conjugation.
Taube and his students have provided first reaction involving electron transfer by inner sphere mechanism. The overall reaction is:
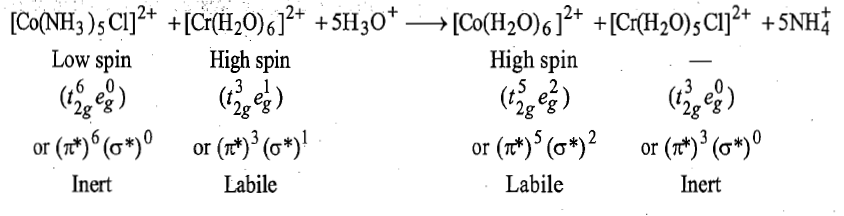
In this reaction, Co3+is reduced by Cr2+ and the bridging ligand Cl- is transferred from coordination sphere of cobalt to that of chromium.
i.
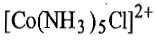
is kinetically inert and Cl- is the bridging ligand.
ii.
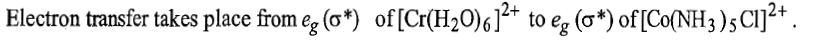
Thus, electron transfer by inner sphere mechanism is fast
The various steps of the mechanism of the reaction are shown below:
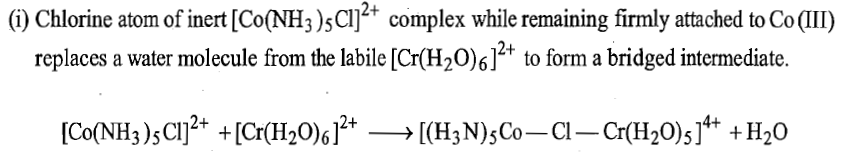
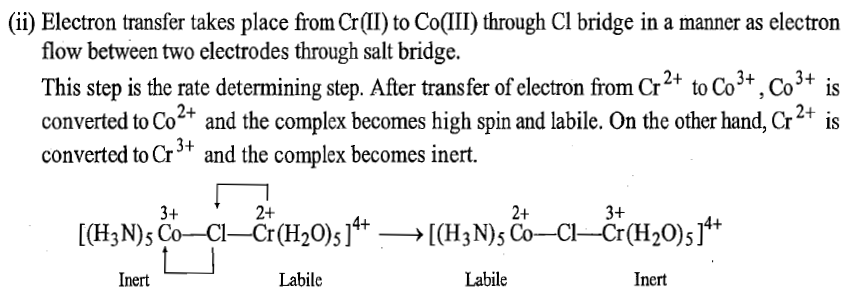
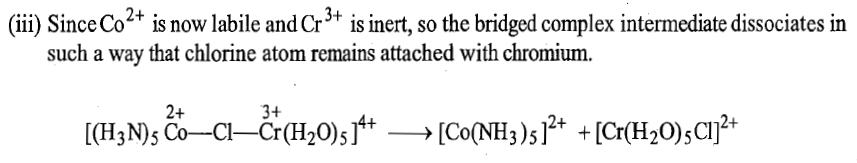

Another example of electron transfer that occurs by inner sphere mechanism without transfer of ligand is:
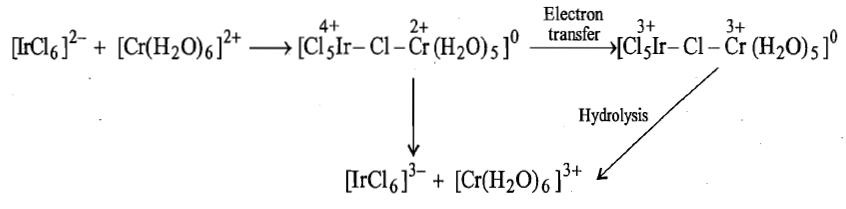
In the bridged complex or precursor complex, after electron transfer, the bond between the reduced metal ion and the bridge [Ir (III)-Cl] is stronger than the bond between the oxidised metal ion and the bridge [Cr(III)-Cl] and the latter will be broken before the former. Thus, in this reaction,
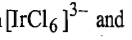
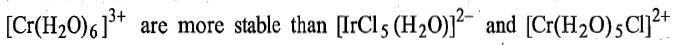
respectively, the other possible products. This reaction indicates that ligand transfer is not a requirement of the inner sphere mechanism.
Outer Sphere mechanism
In this type of reaction, both complexes participating in the reaction undergo substitution reactions more slowly than the rate of electron transfer. The oxidant and the reductant come as close to each other as possible, and the coordination spheres stay intact. The transfer of an electron takes place from the reductant to the oxidant. Thus, an outer sphere mechanism involves electron transfer from the reductant to the oxidant when the intact coordination spheres are in contact at their outer edges, i.e., the distance between two metals is minimal.
An outer sphere electron transfer may occur in the following elementary steps:
In the first step, the oxidant and reductant come closer and form a precursor complex:

In the final step, the ion pair is dissociated into products:


In option d,
Two species are involved: [CrO4]2- (chromate ion) and [Fe(CN)6]4- (hexacyanoferrate(II) ion). This reaction involves electron transfer, and it appears to be a redox reaction where one species loses electrons (oxidation) and the other gains electrons (reduction).
To determine which species is the reductant (donating electrons) and which is the oxidant (accepting electrons), we can analyze the oxidation states of the elements involved.
In the chromate ion, the oxidation state of chromium (Cr) is +6, and in the hexacyanoferrate(II) ion, the oxidation state of iron (Fe) is +2.
When a species undergoes oxidation, its oxidation state increases, while in reduction, the oxidation state decreases. In this case, we can see that the oxidation state of chromium increases from +6 to a higher value, indicating that it is being oxidized. Conversely, the oxidation state of iron decreases from +2 to a lower value, indicating that it is being reduced. In the context of electron transfer reactions, the reductant is the species that donates electrons, and the oxidant is the species that accepts electrons. In this reaction, the [Fe(CN)6]4- complex is the reducing agent, meaning it donates electrons while the [CrO4]2- complex is the oxidizing agent-meaning it accepts electrons. However, the chromium complex is not a metal complex ion in the traditional sense. it is an oxyanion of chromium (a chromate ion).