Correct option is D
The free gas and the adsorbed gas are in dynamic equilibrium, and the fractional coverage of the surface depends on the pressure of the overlying gas. The variation of θ with pressure at a chosen temperature is called the adsorption isotherm.
The Langmuir isotherm
The simplest physically plausible isotherm is based on three assumptions:
1. Adsorption cannot proceed beyond monolayer coverage.
2. All sites are equivalent and the surface is uniform (that is, the surface is perfectly flat on a microscopic scale).
3. The ability of a molecule to adsorb at a given site is independent of the occupation of neighbouring sites (that is, there are no interactions between adsorbed molecules).

For adsorption with dissociation, the rate of adsorption is proportional to the pressure and to the probability that both atoms will find sites, which is proportional to the square of the number of vacant sites,
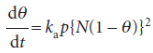
The rate of desorption is proportional to the frequency of encounters of atoms on the surface, and is therefore second-order in the number of atoms present:
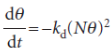
The condition for no net change leads to the isotherm
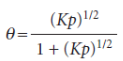
The surface coverage now depends more weakly on pressure than for non-dissociative adsorption.













