Correct option is B
In current terminology, the structurally analogous fragments are said to be isolobal, and the relationship is expressed by the symbol

The origin of the name is the lobe-like shape of a hybrid orbital in a molecular fragment. Two fragments are isolobal if their highest energy orbitals have the same symmetry (such as the σ symmetry of the H1s and a Csp3 hybrid orbital), similar energies, and the same electron occupation (one in each case in H1s and Csp3). Table lists some selected isolobal fragments and the first line shows isolobal fragments with a single frontier orbital. The recognition of this family permits us to anticipate by analogy with H−H that molecules such as H3C−CH3 and (OC)5Mn−CH3 can be formed. The second line of Table lists some isolobal fragments with two frontier orbitals, and the third line lists some with three.
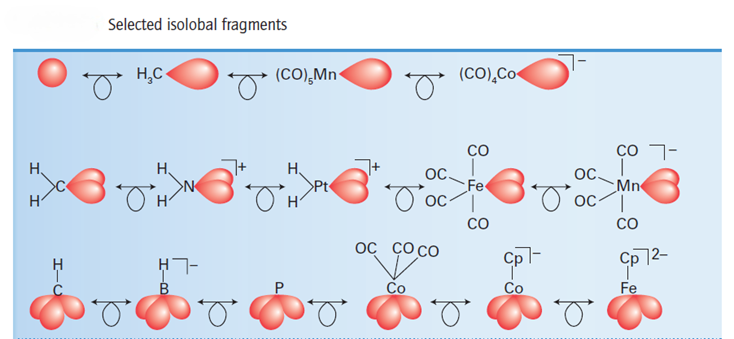
Using the isolobal principle, one can relate clusters that contain fragments having analogous orbital properties. Some isolobal pairs of metal carbonyl and hydrocarbon fragments are:
i. CH3: Carbon has four valence electrons
Three electrons are given by three H atoms. Total electrons:4+3=7
[Fe (CO)5]+: Electronic configuration of Fe: [Ar] 3d⁶4s²
CO donates two electrons. Total electrons: 8+ (5×2)-1 = 17
Positive charge leads to removal of one electron.
ii. [CH3]+: Carbon has four valence electrons
Three electrons are given by three H atoms. Total electrons: 4+3-1=6
[Cr(CO)5]-: Electronic configuration of Cr: [Ar] 3d⁵4s¹.
Total electrons: 6+(5×2)+1=17
Negative charge leads to addition of one electron.
iii. [CH3]+: Carbon has four valence electrons
Three electrons are given by three H atoms. Total electrons: 4+3-1=6
[Ni(CO)3]: Electronic configuration of Ni :[Ar] 3d⁸4s²
Total electrons: 10+(3×2) = 16
iv. [CH]+:Carbon has four valence electrons
One electron is given by one H atom. Total electrons: 4+1-1=4
[Co(Cp)] : Electronic configuration of Co: [Ar] 3d⁷4s².
Cyclopentadienyl ligand (Cp:C5H5-) donates five electrons. Total electrons: 9+5=14







