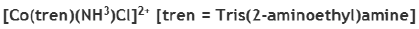Correct option is A
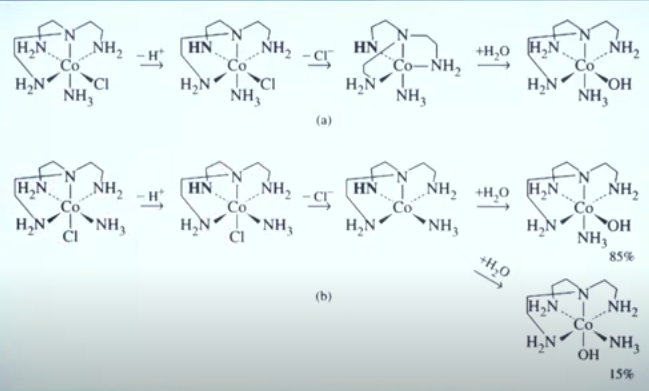
Reactions with [Co(tren)(NH3)CI]2+isomers show that the nitrogen atom trans to leaving group is most likely to be deprotonated in the conjugate base mechanism. The dominant kinetic product in both reactions is hypothesized to proceed by a trigonal-bipyramidal intermediate or transition state with the amido ligand in the trigonal plane. The reaction in Figure a has a much lower barrier to trigonal-bipyramidal geometry (with only slightly widening of two N-Co-N than does the Figure b reaction, which requires more dramatic rearrangement square-pyramidal structure. The activation energy associated with hydrolysis of the square pyramidal intermediate in Figure b, where the hydroxo ligand would engage an oxygen orthogonal to the amido nitrogen, is sufficiently high that the (slow) rearrangement trigonal-bipyramidal structure, followed by hydrolysis is still the faster pathway.