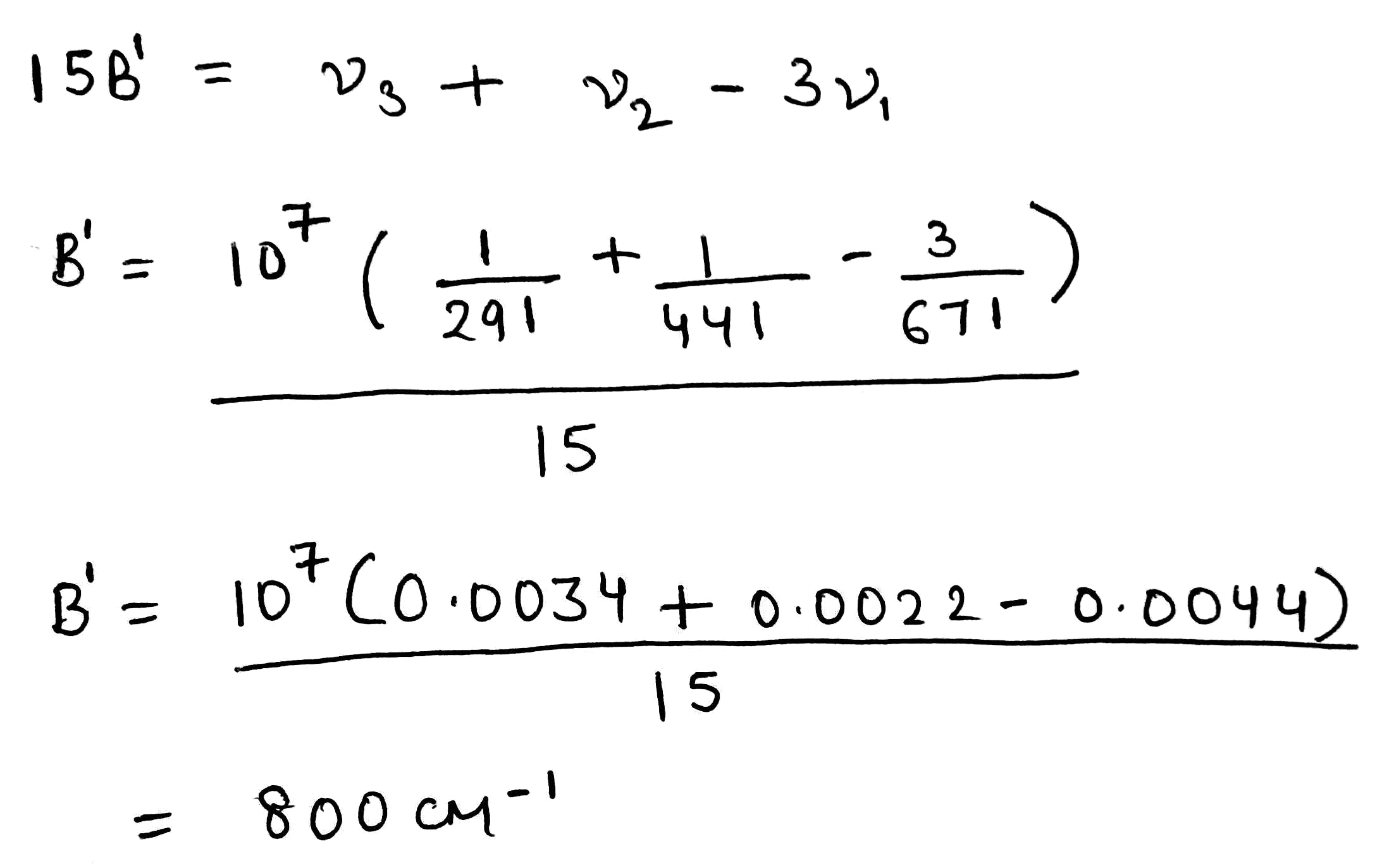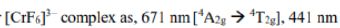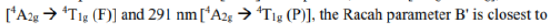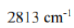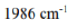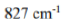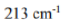Correct option is C
In d3octahedral complexes the d-orbitals split into
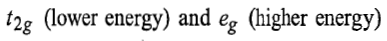
orbital. Therefore, in ground state the electronic configuration is
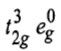
and there are two holes in eg orbitals.
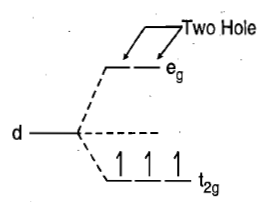
There is only one electronic arrangement corresponding to this electronic configuration
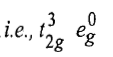
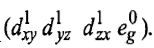
Therefore, this arrangement is electronically non-degenerate and can be represented as
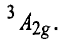
This case is similar to d8 octahedral complex. When an electron is excited to any one of the eg orbitals, the electronic configuration becomes
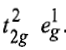
Now there are two holes, one in t2g and one in eg orbitals. There are two triply degenerate arrangements of holes corresponding to this electronic configuration. The lower energy state will arise when two holes occupy the orbitals as far apart as possible. Therefore, the two holes may be present either in
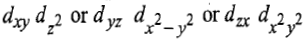
Thus, this state is triply degenerate and is represented as
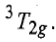

Therefore, there are three transitions of holes from ground states to three other excited states. Transition of holes is similar to electronic transition.

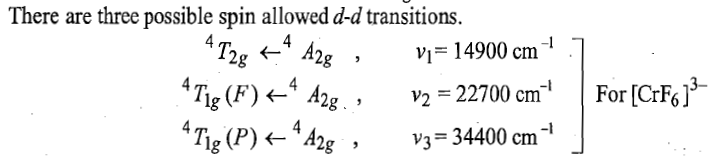
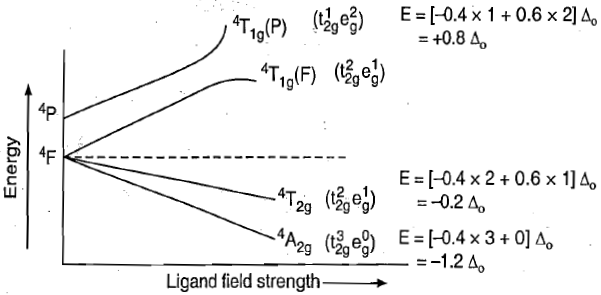
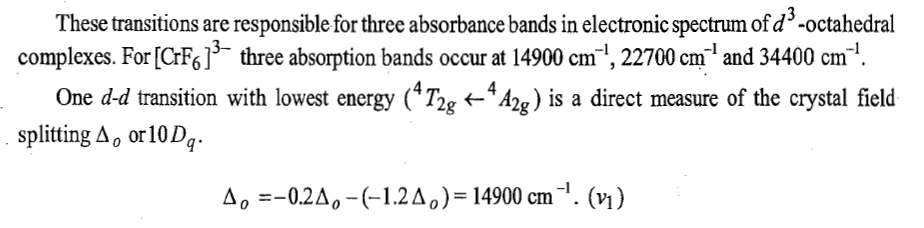
Racah parameter
Different terms of a configuration have different energies due to inter-electronic repulsion. The inter-electronic repulsion (or mixing) which causes the bending of the lines is expressed in terms of the Racah parameters B and C which can be calculated from linear combinations of coulomb and exchange integrals. These parameters are empirical quantities and are obtained from the spectra of free ions in gas phase. For the terms of same spin multiplicity, the difference in energy is only the function of B. For example, the difference in energy between free ion ground state term F and an excited state term P of same multiplicity for d2,d3,d7 and d8-metal ions is 15 B. However, both parameters B and C are necessary for terms with different multiplicities, for example, in the Cr3+ (d3) ion the difference between energy between 4F and 2G is 4B + 3C. For most transition metal ions value of B lie in the range of 700 - 1100 cm-1. C is equal to ~ 4B.
The value of B, labeled as B' for a complex is always less than that of the free ion because of delocalization of metal electrons in molecular orbitals that encompass both the metal and ligands. Due to delocalization of electrons inter-electronic repulsion is decreased. The delocalization increases the average separation of d- electrons and hence reduces their mutual repulsion. The reduction of B from its free ion value is called nephelauxetic effect or cloud expanding effect. The nephelauxetic effect is represented in terms of nephelauxetic parameter (β) which is given by: