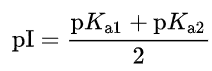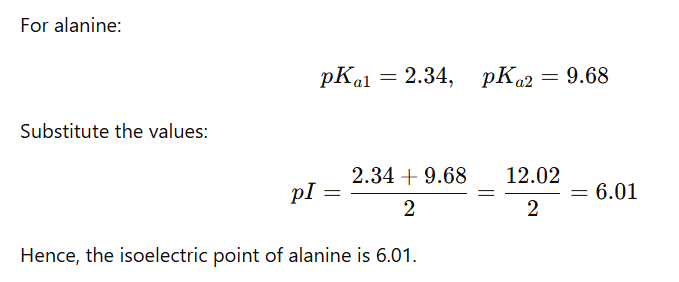Correct option is A
The isoelectric point , is the pH at which a molecule carries no net electrical charge or is electrically neutral in the statistical mean. The net charge on the molecule is affected by pH of its surrounding environment and can become more positively or negatively charged due to the gain or loss, respectively, of protons (H+).
Biological amphoteric molecules such as proteins contain both acidic and basic functional groups. Amino acids that make up proteins may be positive, negative, neutral, or polar in nature, and together give a protein its overall charge. At a pH below their pI, proteins carry a net positive charge; above their pI they carry a net negative charge.
For an amino acid with only one amine and one carboxyl group, the pI can be calculated from the mean of the pKas of this molecule.