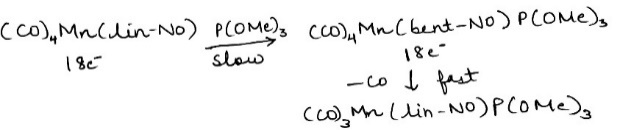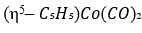Correct option is B
Associative substitution describes a pathway by which compounds interchange ligands. Associative pathways are characterized by binding of the attacking nucleophile to give a discrete, detectable intermediate followed by loss of another ligand. Associative substitution differs from dissociative in that the incoming ligand binds to the complex before the departing ligand leaves. This is typical of 16e- complexes because the intermediate is then 18e- and is analogous to the associative SN2 organic reaction. Complexes that undergo associative substitution are either coordinatively unsaturated or contain a ligand that can change its bonding to the metal, e.g. change in hapticity or bending of a nitrogen oxide ligand (NO).
In special situations, some ligands participate in substitution reactions leading to associative pathways. These ligands can adopt multiple motifs for binding to the metal, each of which involves a different number of electrons "donated." Nitric oxide typically binds to metals to make a linear MNO arrangement, wherein the nitrogen oxide is said to donate 3e- to the metal. In the course of substitution reactions, the MNO unit can bend, converting the 3e- linear NO ligand to a 1e- bent NO ligand.
P(OMe)3 is a two-electron donor.