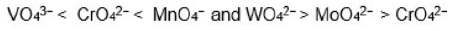Correct option is A
Charge transfer transition is the transfer of an electron between orbitals that are centred on different atoms. Unlike d-d transitions, charge transfer transitions are Laporte and spin allowed that is
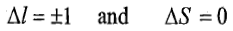
Thus, charge transfer transitions give rise to more intense absorptions. When these transitions occur in visible region, the compound show intense colour. A charge transfer transition may be regarded as an internal redox process.
If the transfer of an electron takes place from the ligand to metal, then charge transfer is called ligand to metal charge transfer.
Conditions for LMCT:
i. Metal should be in high oxidation state so that it has high ionization energy, smaller size and vacant orbitals at low energies.
ii. Ligands should have lone pair of electrons of relatively high energy and low electron affinity.

Energy required to transfer an electron from ligand to metal depends upon the lowest unoccupied molecular orbital (LUMO) of the metal cation and the highest occupied molecular orbital (HOMO) of the ligand.
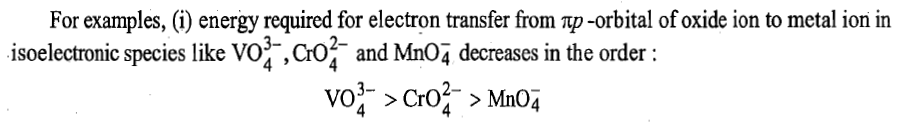
On moving from
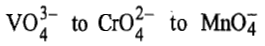
size of metal cation decreases in the order
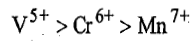
because in this direction effective nuclear charge increases and energy of acceptor orbitals (d-orbitals) decreases. Therefore, energy of charge transfer decreases.

On moving from 3d to 4d to 5d-series of transition metals in a group, for a given oxidation state, size of metal cation increases and energy of LUMO ( that is vacant d orbitals) centred on metal cation increases and therefore, energy required for transfer of an electron from HOMO of oxide ion to LUMO of metal cation in tetra oxo-anions increases on moving down the group.
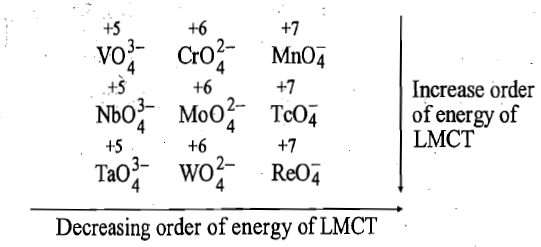
Tetra oxo-anions of 4d and 5d-series transition metals are colorless because energy difference between 2p of oxide ion and 4d and 5d orbitals of transition metals is very large and electron transfer from pi-p orbitals of oxide to d orbitals of metal requires a high energy which lies in UV-region.
Additionally, using equation

Photon energy is the energy carried by a single photon. The amount of energy is directly proportional to the photon's electromagnetic frequency and thus, equivalently, is inversely proportional to the wavelength. The higher the photon's frequency, the higher its energy. Equivalently, the longer the photon's wavelength, the lower its energy.