Correct option is B
Iron pentacarbonyl, also known as iron carbonyl, is the compound with formula Fe(CO)5. Most metal carbonyls have 18 valence electrons, and Fe(CO)5 fits this pattern with 8 valence electrons on Fe and five pairs of electrons provided by the CO ligands. Fe(CO)5 adopts a trigonal bipyramidal structure with the Fe atom surrounded by five CO ligands: three in equatorial positions and two axially bound.
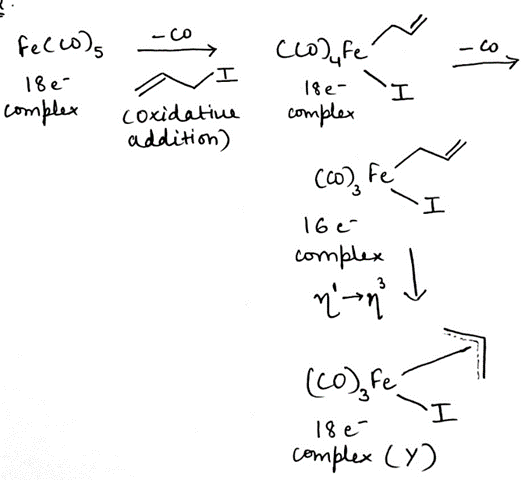
For transition metals, oxidative reaction results in the decrease in the dn to a configuration with fewer electrons, often 2e fewer. Oxidative addition is favored for metals that are (i) basic and/or (ii) easily oxidized. Metals with a relatively low oxidation state often satisfy one of these requirements, but even high oxidation state metals undergo oxidative addition.
In classical organometallic chemistry, the formal oxidation state of the metal and the electron count of the complex both increase by two. One-electron changes are also possible and in fact some oxidative addition reactions proceed via series of 1e changes. Although oxidative additions can occur with the insertion of a metal into many different substrates, oxidative additions are most commonly seen with H–H, H–X, and C–X bonds because these substrates are most relevant to commercial applications.
Oxidative addition requires that the metal complex have a vacant coordination site. For this reason, oxidative additions are common for four- and five-coordinate complexes.
Potential ligands that do not have a lone pair or filled π type orbital are still able to interact with transition metal complexes but only by breaking a σ bond. This is the first step in a wide variety of processes and is described as oxidative addition because the formal oxidation state of the transition metal is raised by two, for example, M (0) to M(II), in the process. This is the result of having two extra ligands bearing a formal negative charge.
The number of coordinated ligands also increases by two so the starting complex is usually in low oxidation state (0 or 1; the diagram shows 0) and coordinatively unsaturated, that is, it has an empty site for a ligand whereas the product is usually coordinatively saturated, that is, it cannot accept another ligand unless it loses one first.
The number of atoms involved in bonding to the metal is shown by the hapto number η. The metal can either form a σ bond to a single carbon (hence η1), or form a π complex with the p orbitals of all three carbons of the allyl system and this would be η3. The distinction is very important for electron counting as these two different situations contribute 1 and 3 electrons, respectively, to the complex.
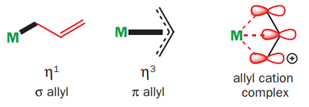
Fe ([Ar] 3d⁶4s²) donates eight electrons, CO donates two electrons, I- donates one electron
Additional Knowledge
In chemistry, a pentagonal bipyramid is a molecular geometry with one atom at the centre with seven ligands at the corners of a pentagonal bipyramid.







