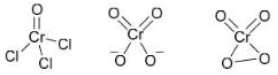
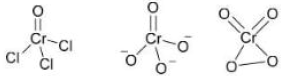
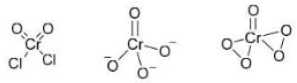
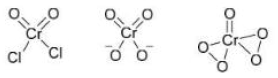
Correct option is D
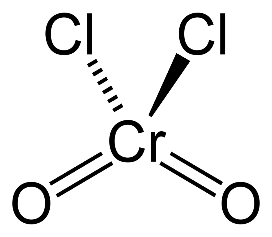
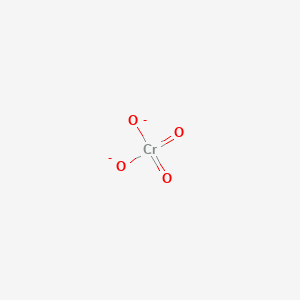
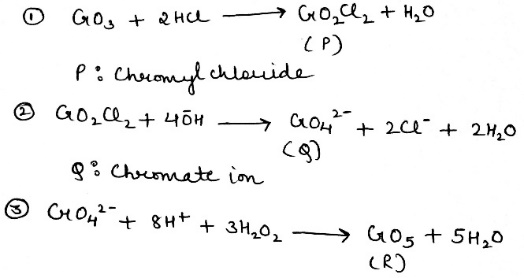
When CrO₃ (chromium trioxide) is exposed to HCl gas, it forms chromyl chloride (CrO₂Cl₂) , which is a red vapor. This reaction is:
When P (CrO₂Cl₂) is passed through a dilute solution of NaOH , a yellow complex ion is formed.
When an acidified solution of H₂O₂ is added to the solution of Q (CrO₄²⁻) , a dark blue compound is formed.