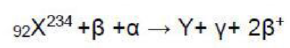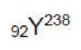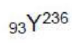Correct option is B
Beta decay (β-decay) is a type of radioactive decay in which an atomic nucleus emits a beta particle (fast energetic electron or positron), transforming into an isobar of that nuclide.
The two types of beta decay are known as beta minus and beta plus. In beta minus (β-) decay, a neutron is converted to a proton, and the process creates an electron and an electron antineutrino; while in beta plus (β+) decay, a proton is converted to a neutron and the process creates a positron and an electron neutrino. β+decay is also known as positron emission.
An example of electron emission (β-decay) is the decay of carbon-14 into nitrogen-14 with a half-life of about 5,730 years:
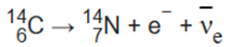
In this form of decay, the original element becomes a new chemical element in a process known as nuclear transmutation. This new element has an unchanged mass number A, but an atomic number Z that is increased by one.
An example of positron emission (β+ decay) is the decay of magnesium-23 into sodium-23 with a half-life of about 11.3 s:
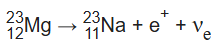
β+ decay also results in nuclear transmutation, with the daughter element having an atomic number that is decreased by one.
Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or "decays" into a different atomic nucleus, with a mass number that is reduced by four and an atomic number that is reduced by two. While alpha particles have a charge +2 e, this is not usually shown because a nuclear equation describes a nuclear reaction without considering the electrons – a convention that does not imply that the nuclei necessarily occur in neutral atoms.
A gamma ray, also known as gamma radiation (symbol γ ), is a penetrating form of electromagnetic radiation arising from high energy interactions like the radioactive decay of atomic nuclei or astronomical events like solar flares. One example of gamma ray production due to radionuclide decay is the decay scheme for cobalt-60. First, 60Co decays to excited 60Ni by beta decay emission of an electron of 0.31 MeV. Then the excited 60Ni decays to the ground state by emitting gamma rays in succession of 1.17 MeV followed by 1.33 MeV. This path is followed 99.88% of the time:
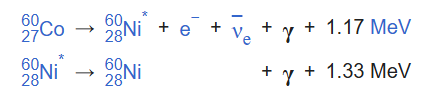
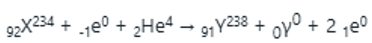
When
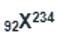
nuclei collides with

the mass number and atomic number of the resulting nuclei will be 238 and 93 respectively.
After the collision, Y and

are formed. The release of two
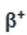
(positron) particles will decrease the atomic number by two units whereas the mass number will be unchanged. The atomic number will be 93-2=91.