Correct option is A
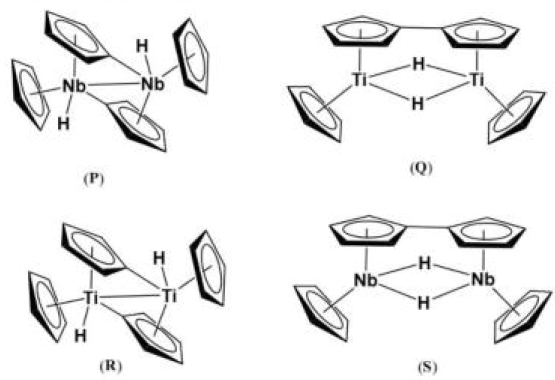
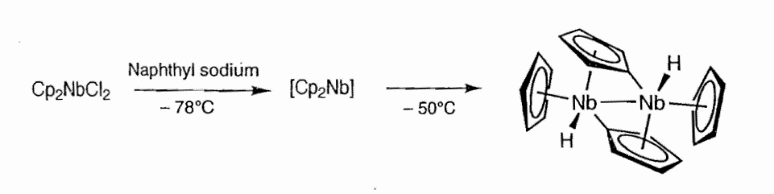
Cp2Nb is a 15e- complex with a highly reactive Nb([Kr] 4d⁴5s¹) with a d3 configuration. It is highly unstable and exists in the monomeric form only below -78°C. Above -50°C, the molecule dimerises to give an unusual structure. Two molecules of niobocene react with one another to produce the structure shown. Note that two of the Cp rings are dianionic forming both a traditional anionic η5-π-type donor to one metal, while bridging and acting as an anionic le- σ-donor to the other metal centre.
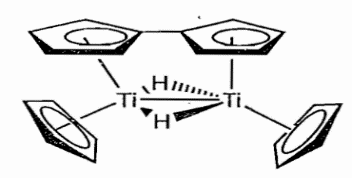
Cp2Ti is a 14e- complex with a highly reactive d2 electronic configuration. Two molecules of titanocene also react with one another to produce a bimetallic complex that may well look just like the niobium complex discussed above. But it undergoes a further reaction (perhaps due to steric crowding) leading to the coupling of two σ bound Cps to produce C-C bound bis-Cp and the complex shown below.