Correct option is C
Electron paramagnetic resonance (EPR) or electron spin resonance (ESR) spectroscopy is a method for studying materials that have unpaired electrons. The basic concepts of EPR are analogous to those of nuclear magnetic resonance (NMR), but the spins excited are those of the electrons instead of the atomic nuclei. EPR spectroscopy is particularly useful for studying metal complexes and organic radicals.
Selection rule for transition in EPR spectroscopy is:
Statement a is correct
(b) Incorrect.
(c) Correct. The energy required for the transition from one spin state to another is proportional to the applied magnetic field. The relation can be written as:
g is the g-factor which is an equivalent of chemical shift in NMR, Beta is Bohr's magneton ( 2.274 × 10-24 J T-1), B is the applied magnetic field
g-factor is anisotropic quantity and it has higher value for the axis with higher electron density. g value along z-axis is denoted as g∥.Along x and y axis, it is written as g⊥
In tetragonally elongated Cu (II) complexes (as shown below), the orbital along z-axis are more stable and have higher electron density compared to the orbitals along x and y axis. This leads to positive coupling with g∥
The Jahn–Teller theorem essentially states that any non-linear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species.
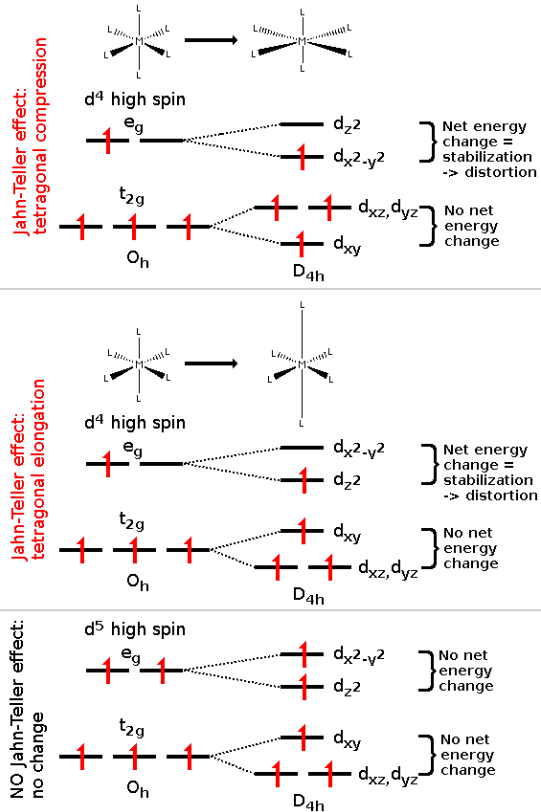
In tetragonally compressed Cu (II) complexes, the ligands along x, y axis are a little far apart, making the d orbitals along x and y axis more stable.











