Correct option is B
Outer sphere mechanism
In this type of reaction, both complexes participating in the reaction undergo substitution reactions more slowly than the rate of electron transfer. The oxidant and the reductant come as close to each other as possible, and the coordination spheres stay intact. The transfer of an electron takes place from the reductant to the oxidant. Thus, an outer sphere mechanism involves electron transfer from the reductant to the oxidant when the intact coordination spheres are in contact at their outer edges, i.e., the distance between two metals is minimal.
An outer sphere electron transfer may occur in the following elementary steps:
In the first step, the oxidant and reductant come closer and form a precursor complex:

In the second step, there is activation of the precursor complex, which includes reorganization of the solvent molecules and changes in M–L bond lengths, occurring before electron transfer. Then, the electron transfer takes place:

In the final step, the ion pair is dissociated into products:

The electron transfer is very fast when both the complexes are low spin including that the electron transfer takes place from t2g (π*) of reductant to the t2g (π*) of oxidant. The first reason is that energy levels of these two t2g orbitals are same. The t2g orbitals are not shielded from the ligands and the electron transfer from and to is easier and no input energy is required. The second reason is that there is no appreciable change in M-L bond length due to π*- π* electron transfer.

When both reactants in a redox reaction are kinetically inert, electron transfer must take place by a tunnelling or outer-sphere mechanism. In a self-exchange reaction, the left- and right-hand sides of the equation are identical. Only electron transfer, and no net chemical reaction, takes place.
Clearly, the reactants must approach closely for the electron to migrate from reductant to oxidant. This reductant–oxidant pair is called the encounter or precursor complex. When electron transfer occurs, there is an important restriction imposed upon it by the Franck–Condon approximation.
According to Frank-Condon principle, electron transfer takes place when the energy levels of the participating orbitals are same and the electron transfer takes place much more rapidly than the change in position of nuclei. Thus, during electron transfer M-L bond lengths remain unchanged.
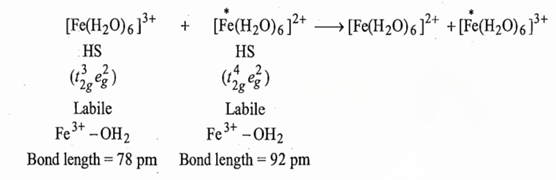
In this reaction an electron is transferred from t2g of Fe (II) to t2g of Fe (III). The Fe2+-OH2 and Fe3+-OH2bond lengths are 92 pm and 78 pm respectively, i.e., Fe3+-OH2 bond length is 14 pm smaller than that of Fe2+-OH2. This indicate that the energy levels of the orbitals are not equal. Since the Fe-O bond length does not change during the electron transfer, this transfer of electron without any input of energy will produce [Fe (H2O)6]3+with Fe-O bond similar to Fe-O bond in [Fe (H2O)6]2+ and the [Fe(H2O)6]2+with Fe-O bond similar to [Fe(H2O)6]3+. These two products are in vibrationally excited sate. Both the products will release energy in the form of heat by vibrating to reach the equilibrium bond length. It seems that energy is created but it is the violation of the first law of thermodynamics. Consequently, an enough vibrational activation energy has been provided to each complex ion to bring them to the same energy levels and then transfer of electron to take place.
Consequently, the electron transfer is most probable when an activation energy has been given to each complex ion. The activation energy causes the shortening of the bonds in [Fe (H2O)6]2+ and lengthening of the bonds in [Fe (H2O)6]3+ until the participating orbitals are of the same energy. In case of self-exchange reaction, the Fe-O bond lengths have reached the same intermediate value. For cross reactions, the M-L bond lengths do not become same values but the orbital energy may be.
eg (σ*) orbital is involved in electron transfer in between [Cr(H2O)6]2+(eg1) and [Cr(H2O)6]3+(eg0).
Outer sphere electron transfer reactions between complexes of different metal cations that is cross reactions are faster than self exchange reactions.
Second order rate constant is given as:








