Correct option is A
In organic chemistry, hydroformylation, also known as oxo synthesis or oxo process, is an industrial process for the production of aldehydes (R−CH=O) from alkenes (R2C=CR2). This chemical reaction entails the net addition of a formyl group (−CHO) and a hydrogen atom to a carbon-carbon double bond.
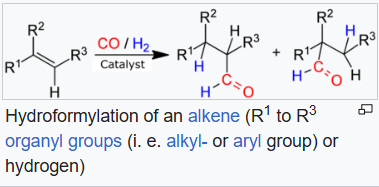
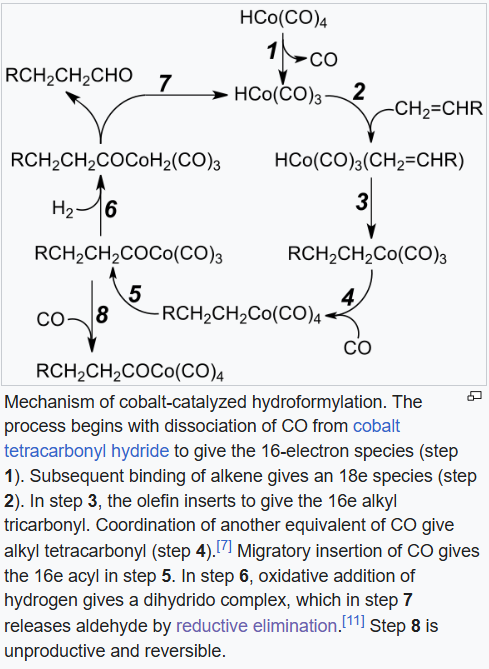
The process entails treatment of an alkene typically with high pressures (between 10 and 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. In one variation, formaldehyde is used in place of synthesis gas. Transition metal catalysts are required. Invariably, the catalyst dissolves in the reaction medium, i.e. hydroformylation is an example of homogeneous catalysis.

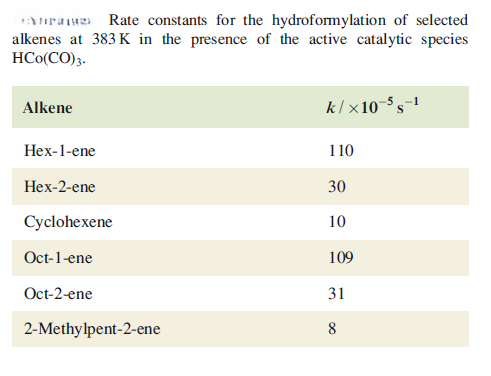
Terminal olefins (olefins with a double bond at the end of the carbon chain) tend to react faster compared to internal olefins (olefins with a double bond somewhere in the middle of the carbon chain). This can be attributed to stearic effects.
Asymmetric hydroformylations are affected by reaction temperature and CO pressure, either or both potentially altering reaction kinetics. As an example, in asymmetric hydroformylation of styrene, using higher CO pressures favors the formation of branched isomers, whereas lower CO pressure results in linear isomers.