Correct option is A
The Jahn–Teller theorem states that any non-linear molecular system in a degenerate electronic state will be unstable and will undergo distortion to form a system of lower symmetry and lower energy, thereby removing the degeneracy.
Octahedral complexes of d9 and high-spin d4 ions are often distorted, e.g. CuF2 and [Cr(OH2)6]2+ , so that two metal–ligand bonds (axial) are different lengths from the remaining four (equatorial). This is shown in structures (elongated octahedron) and (compressed octahedron). For a high-spin d4 ion, one of the eg orbitals contains one electron while the other is vacant. If the singly occupied orbital is the dz2 , most of the electron density in this orbital will be concentrated between the cation and the two ligands on the z axis. Thus, there will be greater electrostatic repulsion associated with these ligands than with the other four, and therefore the complex suffers elongation. Conversely, occupation of the dx2-y2 orbital would lead to elongation along the x and y axes. A similar argument can be put forward for the d9 configuration in which the two orbitals in the eg set are occupied by one and two electrons respectively. Electron-density measurements confirm that the electronic configuration of the Cr2+ ion in [Cr(OH2)6]2+ is approximately dxy1dyz1dxz1dz2(1). The corresponding effect when the t2g set is unequally occupied is expected to be very much smaller since the orbitals are not pointing directly at the ligands. This expectation is usually, but not invariably, confirmed experimentally. Distortions of this kind are called Jahn–Teller or tetragonal distortions.
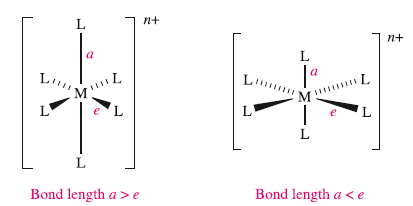
In octahedral complexes, the Jahn–Teller effect is most pronounced when an odd number of electrons occupy the eg orbitals.
w: weak Jahn–Teller effect (t2g orbitals unevenly occupied)
s: strong Jahn–Teller effect expected (eg orbitals unevenly occupied)
blank: no Jahn–Teller effect expected.
A. H2O is a weak field ligand, thus it will form a high spin complex. Mn has an oxidation state of +3. The electronic configuration of Mn3+ is 3d4. Electrons are unsymmetrically filled in eg orbital.
B. H2O is a weak field ligand, thus it will form a high spin complex. Ni has an oxidation state of +2. The electronic configuration of Ni2+ is 3d8. Electrons are symmetrically filled.
C. In VCl4 molecule, V has an oxidation state of +4. The electronic configuration of V4+ is 3d1. Electrons are unsymmetrically filled.