Correct option is B
The isomers in which the same types and number of ligands are coordinated to the metal atom or cation but with different spatial arrangements are called stereoisomers. In other words, stereoisomers are isomers that differ only in the spatial arrangement of ligands coordinated to metal cation or atom. Stereoisomerism is classified into two types :
(1) Geometrical isomerism
(2) Optical isomerism
Geometrical isomerism
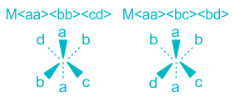
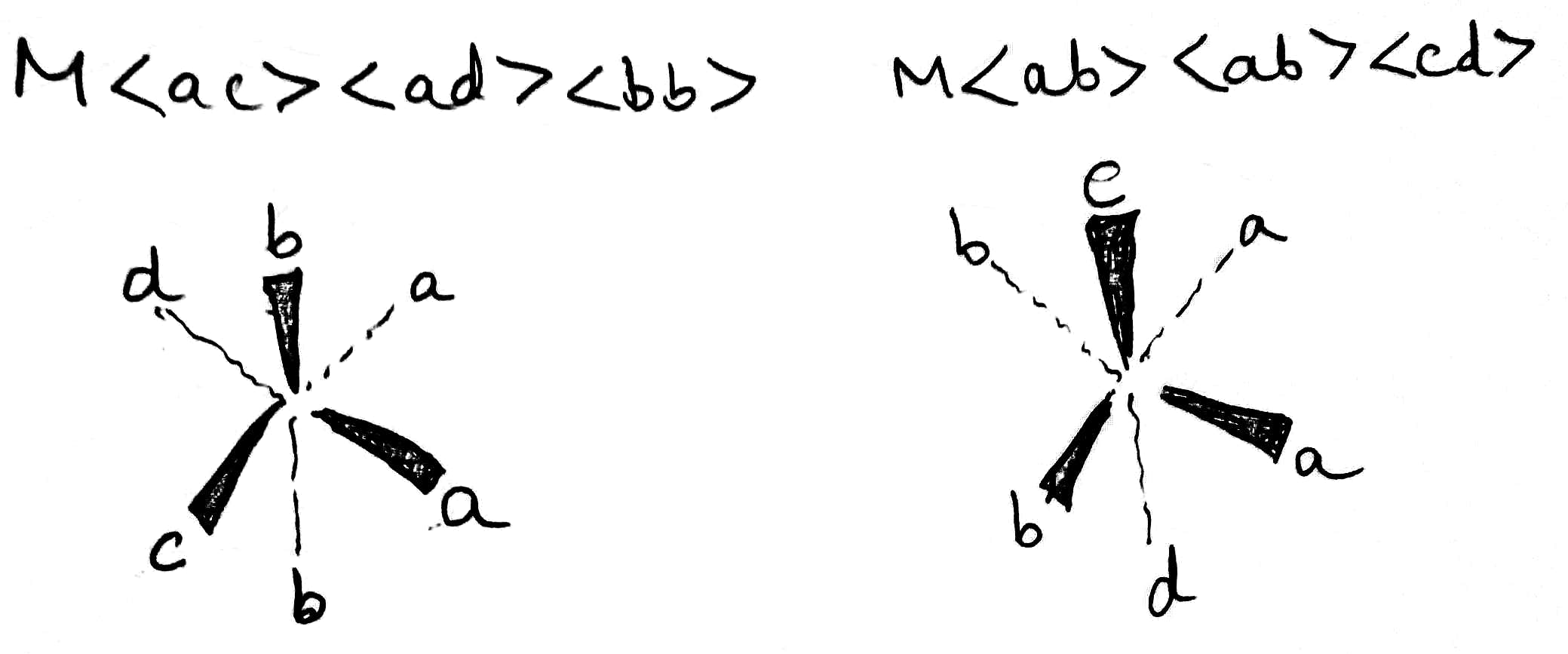
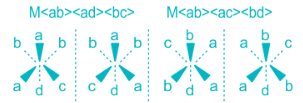
Stereoisomers in which the relative positions or orientations of the ligands or more specifically donor atoms around the metal cation is different are called geometrical isomers and this phenomenon is called geometrical isomerism. Geometrical isomers can not be inter-converted without breaking of M-L bonds. Geometrical isomers exists only in pairs, in one isomer the two particular ligands are adjacent to each other and in the other the two are in opposite sides in the structural formula. Thus, the isomer in which two particular ligands (either identical or non-identical) occupy the adjacent positions of each other is called cis-isomer and the isomer in which two particular ligands occupy opposite positions to each other is called trans-isomer (The Latin word cis means next to, trans means across). cis- and trans- isomers are different compounds with different properties like melting points, dipole moments, solubility, colours and chemical properties. Geometrical isomerism is most common in complexes having coordination number of 4 and 6.
Optical isomerism
Optical isomers or enantiomers are pairs of molecules or ions which are non-superimposable mirror images of each other. The term superimposable means that if one structure is laid over the other of the same molecule, the positions of all the atoms will match and the term non-superimposable means that if one structure is laid over the other of the same molecule, the positions of all the atoms will not match. The optical isomers have handedness and are said to chiral because like left and right hands, chiral molecules are non-superimposable. Isomers that are superimposable (i.e., that lack handedness) with their mirror images are said to be non-chiral or achiral. The essential condition for a substance to be chiral (or optically active) is that the substance has no plane of symmetry. If a substance has a plane of symmetry, then it will be achiral (or optically inactive). The substances having no plane of symmetry (or mirror plane symmetry) are always non-superimposable on their mirror images.
Only cis isomers are optically active because there is no plane of symmetry present.
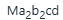
has eight isomers. There are two pairs of enantiomers.