Correct option is A



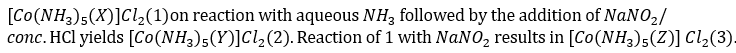
Complex 2 shows two IR spectral bands at 1310 and 1430 cm-1, whereas complex 3 shows the same at 1065 and 1470 cm-1. X, Y and Z, respectively, are



Consider following statements regarding the magnetic moment of the 3d transition metal complexes:
A. Octahedral complexes of (high spin) and (high spin) have approximately equal to their values.
B. Tetrahedral complexes of and octahedral complexes of have orbital contribution added to their magnetic moments.
C. Orbital contribution is quenched for metal ions having A or E ground state.
D. Octahedral complexes of have value at 300 K due to orbital contribution added to their magnetic moments
Choose the correct answer from the options given below:
The possible terms arising from a p1d1 configuration are
For octahedral geometry the triad with no net gain in CFSE, when the ligand field changes from weak to strong, is:
The correct statement for the CO and ligands in the complexes trans – and is:
The correct pair among the following, in which each species shows orbital contribution to its magnetic moment is
The red colour of the gem, ruby is predominantly due to
The value of magnetic moment will be independent of temperature for
(acac=acetylacetonato; OAc=acetate; o-phen=o-phenanthroline; Pz=pyrazolyl)
The number of expected electronic transitions in [Cr(en)3]3+ and trans-[Cr(en)2F2]+ at 4 K is, respectively (en=ethylenediamine)
The products A and B for the given reaction

are, respectively

