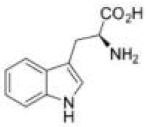
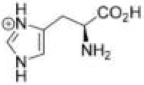
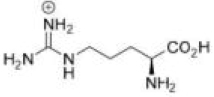
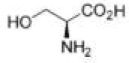
Correct option is C
Proteins have specific amino acids that interact with various molecules or ions. The interaction with [HPO4]2- typically occurs through electrostatic interactions and hydrogen bonding. The amino acids involved must possess functional groups capable of recognizing negatively charged ions like phosphate.
Electrostatic Interactions: Positively charged amino acids can interact with negatively charged phosphate ions. These interactions are crucial in phosphate binding, where a positively charged side chain will stabilize the interaction.
Hydrogen Bonding: Amino acids with hydrogen-bond-donating capabilities can interact with the oxygen atoms in the phosphate group, further stabilizing the interaction. Functional groups capable of donating or accepting hydrogen bonds are important for this interaction.
Side Chain Functionalities: Amino acids like lysine (Lys) and arginine (Arg) possess positively charged side chains that are capable of interacting with negatively charged groups like phosphate due to their protonated amine groups.
In option c, Arginine (Arg) is the amino acid that can effectively recognize [HPO4]2-.Arginine has a positively charged guanidinium group, which can interact electrostatically with the negatively charged phosphate group. Additionally, it can form hydrogen bonds with the oxygen atoms of the phosphate group, stabilizing the binding.