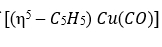Correct option is A
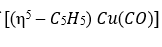
:
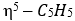
donates five electrons, Cu [[Ar] 4s¹3d¹⁰] donates eleven electrons and CO donates two electrons. Therefore, total electron count is 5+11+2=18 electrons.
In majority of nitrosyl complexes the MNO unit is linear. When NO acts as 3e- donor, the M-NO unit is linear. In the linear structure, NO transfers one electron to metal resulting in decrease in oxidation state of metal and formation of NO+ ion.
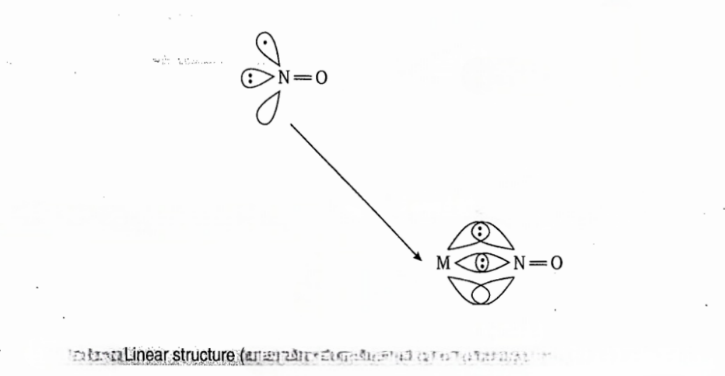
In covalent model, there is no need of rehybridization on N atom of NO because the metal has a singly occupied dπ orbital to overlap with singly occupied π* orbital of NO to form M-Nπ bond and N donates its lone pair of electrons to the empty dσ orbital of metal to form a σ bond.
a.
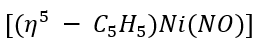
: Ni [[Ar] 3d⁸4s²] is a ten-electron donor. Total electron count in the given complex is: 5+10+3=18
b.
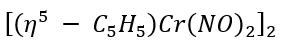
: Cr [[Ar] 3d⁵4s¹] is a six-electron donor. Total electron count in the given complex is 2[5+6+3(2)] =2×17=34
c.
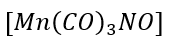
: Mn [[Ar] 4s²3d⁵] is a seven-electron donor. Total electron count in the given complex is 7+2(3) +3=16
d.
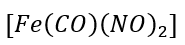
: Fe [[Ar] 3d⁶4s²] is a eight electron donor. Total electron count in the given complex is 8+2+3(2)=16