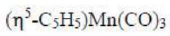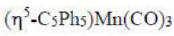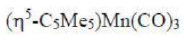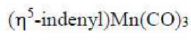Correct option is D
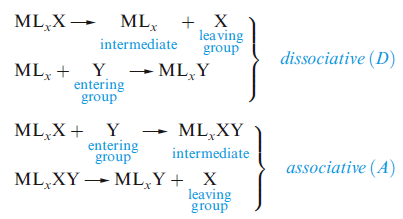
In inorganic substitutions, the limiting mechanisms are dissociative (D), in which the intermediate has a lower coordination number than the starting complex, and associative (A), in which the intermediate has a higher coordination number.
The substitution of a CO ligand by another 2-electron donor (e.g. PR3) may occur by photochemical or thermal activation, either by direct reaction of the metal carbonyl and incoming ligand, or by first replacing a CO by a more labile ligand such as THF or MeCN.
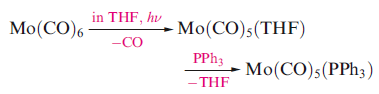
The substitution steps are dissociative. The outgoing ligand leaves, creating a 16-electron metal centre which is coordinatively unsaturated. The entry of a new 2-electron ligand restores the 18-electron count.
In the given question, all the metal complexes contain the metal atom Mn and 3 CO ligands. In each metal complex, Mn ([Ar] 4s²3d⁵) has zero oxidation state. A CO ligand is a two electron donor. Cyclopentadienyl ligand contributes five electrons. Indenyl ligand contributes five electrons. The total electrons present in each metal complex is =5+7+6 = 18 electrons.
The indenyl effect refers to an explanation for the enhanced rates of substitution exhibited by η5-indenyl complexes vs the related η5-cyclopentadienyl complexes.
Associative substitution occurs by the addition of a ligand to a metal complex followed by dissociation of an original ligand. Associative pathways are not typically seen in 18-electron complexes due to the requisite intermediates having more than 18 electrons associated with the metal atom. 18 electron indenyl complexes; however, have been shown to undergo substitution via associative pathways quite readily. This is attributed to the relative ease of η5 to η3 rearrangement due to stabilization by the arene. This stabilization is responsible for substitution rate enhancements of about 108 for the substitution of indenyl complexes compared to the corresponding cyclopentadienyl complex.
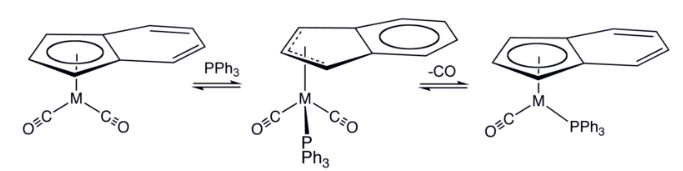
η5 and η3 isomers exist in rapid chemical equilibrium. The rate-limiting step occurs with the attack of the nucleophile on a η3 isomer. The nature of the substituents of the allyl group can strongly affect the kinetics and regiochemistry of the nucleophilic attack.