Correct option is D
Classifying nitrides is not simple, but nearly all nitrides fall into one of the following groups
1.saline nitrides of the group 1 and 2 metals, and aluminium;
2.covalently bonded nitrides of the p-block elements (BN, C2N2, Si3N4, Sn3N4 and S4N4);
3.interstitial nitrides of d-block metals;
4.pernitrides of the group 2 metals.

Thin films of α-BN (which possesses a layer structure) can be deposited by Chemical Vapour Deposition (CVD) using reactions of NH3 with volatile boron compounds such as BCl3 or BF3 at temperatures of ~1000 K.
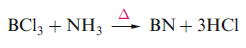
α-Boron nitride films have a range of applications which make use of the material’s hardness, resistance to oxidation and insulating properties.
Aluminium Nitride
Aluminium nitride (AlN) is a solid nitride of aluminium. It has a high thermal conductivity of up to 321 W/(m·K) and is an electrical insulator.
AlN is one of the few materials that have both a wide and direct bandgap and large thermal conductivity. This is due to its small atomic mass, strong interatomic bonds, and simple crystal structure. This property makes AlN attractive for application in high speed and high power communication networks. Many devices handle and manipulate large amounts of energy in small volumes and at high speeds, so due to the electrically insulating nature and high thermal conductivity of AlN, it becomes a potential material for high-power power electronics.

Silicon nitride, Si3N4, has wide applications as a ceramic and refractory material and in the form of whiskers. It is a white, chemically inert amorphous powder, which can be formed by reaction , or by combining Si and N2 above 1650 K.
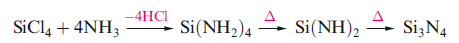
The two most important polymorphs, α- and β-Si3N4, possess 3-dimensional structures containing distorted tetrahedral Si atoms, and 3-coordinate, near-planar N atoms. The detailed crystal structures and the presence of lattice defects have been the subject of debate for many years. A denser, harder polymorph, γ-Si3N4, has been obtained by high pressure, high-temperature (15 GPa, >2000 K) fabrication.
Silicon nitride is often used as an insulator and chemical barrier in manufacturing integrated circuits, to electrically isolate different structures or as an etch mask in bulk micromachining.
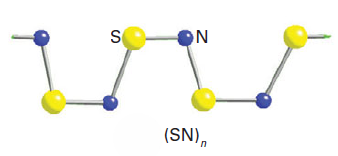
Disulfurdinitride, S2N2, is formed (together with Ag2S and N2) when S4N4 vapour is passed over hot silver wool. It is even more sensitive than its precursor and explodes above room temperature. When allowed to stand at 0oC for several days, disulfurdinitride transforms into a bronze-coloured zig-zag polymer of composition (SN)n, which is much more stable than its precursor, not exploding until 240oC.The compound exhibits metallic conductivity along the chain axis and becomes superconducting below 0.3 K. The discovery of this superconductivity was important because it was the first example of a superconductor that had no metal constituents. Halogenated derivatives have been synthesized that have even higher conductivity. For example, partial bromination of (SN)n produces blue-black single crystals of (SNBr0.4)n, which has room temperature conductivity an order of magnitude greater than (SN)n. Treatment of S4N4 with ICl, IBr, and I2 produces highly conducting, nonstoichiometric polymers with conductivities greater by 16 orders of magnitude than (SN)n.