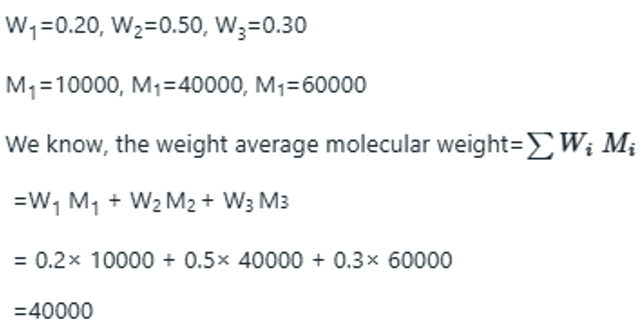Correct option is A
A pure protein is monodisperse, meaning that it has a single, definite molar mass (although there may be small variations, such as one amino acid replacing another, depending on the source of the sample). A synthetic polymer, however, is polydisperse, in the sense that a sample is a mixture of molecules with various chain lengths and molar masses. The various techniques that are used to measure molar masses result in different types of mean values of polydisperse systems.
The weight-average molar mass is the average calculated by weighting the molar masses of the molecules by the mass of each one present in the sample:
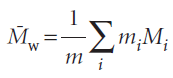
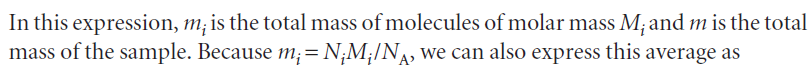
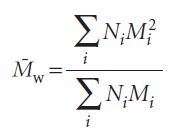
This expression shows that the weight-average molar mass is proportional to the mean square molar mass.