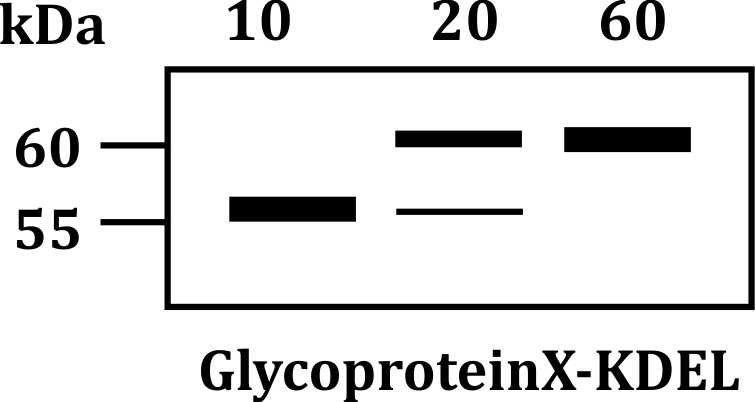Correct option is C
The concept used in this question involves the trafficking of proteins within the cell, particularly focusing on the endoplasmic reticulum (ER) and Golgi apparatus, along with how proteins are modified during their passage through these organelles. Additionally, the presence of the KDEL sequence at the C-terminus of the glycoprotein X is significant, as it serves as a signal for ER retention.
- KDEL Sequence: This sequence ensures the protein is retained in the ER after it is synthesized. However, proteins with KDEL can be transported to the Golgi apparatus for modification before being sent back to the ER.
- Post-translational Modifications: The changes in the molecular weight of the protein observed in the immunoblot are indicative of post-translational modifications, which can occur in the Golgi before the protein is returned to the ER.
Explanation:
In the given experiment, glycoprotein X-KDEL is synthesized and analyzed at 10, 20, and 60 minutes after induction. The appearance of higher molecular weight bands in lanes corresponding to 20 and 60 minutes suggests that the protein undergoes modifications that increase its size.
- KDEL Sequence: This sequence typically causes proteins to be retained in the ER. However, in some cases, these proteins are temporarily transported to the Golgi apparatus where modifications (such as glycosylation) occur.
- The increased molecular weight observed in the immunoblot at 20 and 60 minutes is consistent with modifications in the Golgi, such as the addition of sugar residues, which would add mass to the glycoprotein.
- After these modifications, the protein is likely to be sent back to the ER, where it can then be retrieved if needed. This explains why higher molecular weight forms are observed, as they have undergone modifications before being returned to the ER.
Information Booster:
- KDEL Retention Signal: The KDEL sequence is crucial for keeping certain proteins in the ER. It ensures that proteins involved in the synthesis and folding process are not sent to other organelles like the Golgi or outside the cell.
- Post-translational Modifications: These modifications, which can include glycosylation or phosphorylation, occur in the Golgi apparatus. They are responsible for altering the protein's molecular weight and structure.
- Higher Molecular Weight: The shift to higher molecular weight bands reflects the addition of sugar chains or other modifications that increase the protein's size.
- Golgi Modifications: The Golgi is responsible for modifying proteins by adding or altering carbohydrate groups, which can change their molecular weight.
- ER-Golgi Trafficking: This involves proteins being synthesized in the ER, then sent to the Golgi for further processing before potentially being trafficked back to the ER or secreted.
Additional Information:
- Option 1: The signal sequence is not removed from some of the glycoprotein X-KDEL molecules: Incorrect. If the signal sequence were not removed, the protein would not be processed properly, and we would not see the modifications that increase its size over time. This would show as an unprocessed or immature form of the protein.
- Option 2: Glycoprotein X-KDEL becomes modified in the endoplasmic reticulum after protein synthesis is completed: Incorrect. Although the ER is involved in initial processing and folding, major modifications, such as glycosylation, typically occur in the Golgi, not the ER.
- Option 4: The quality control mechanism in the ER recognizes a pool of glycoprotein X-KDEL as being aberrant and targets it for degradation: Incorrect. If the protein were targeted for degradation, we would see a decrease in the amount of protein over time rather than an increase in molecular weight.