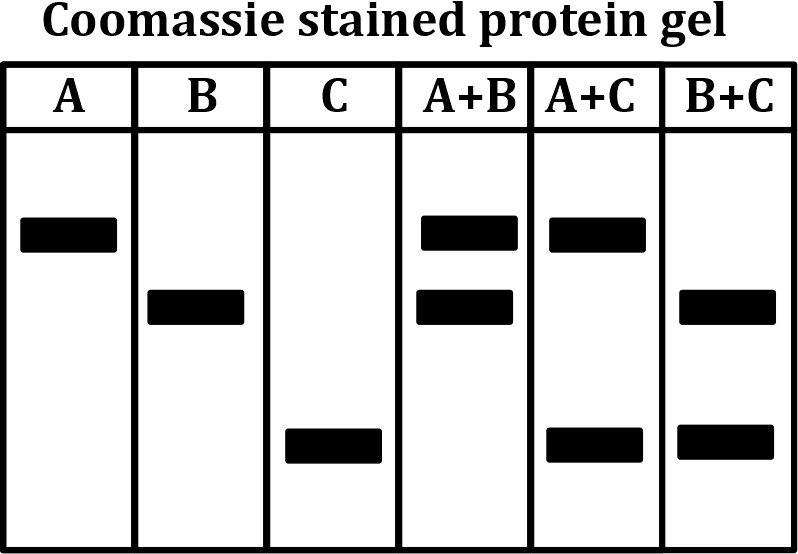
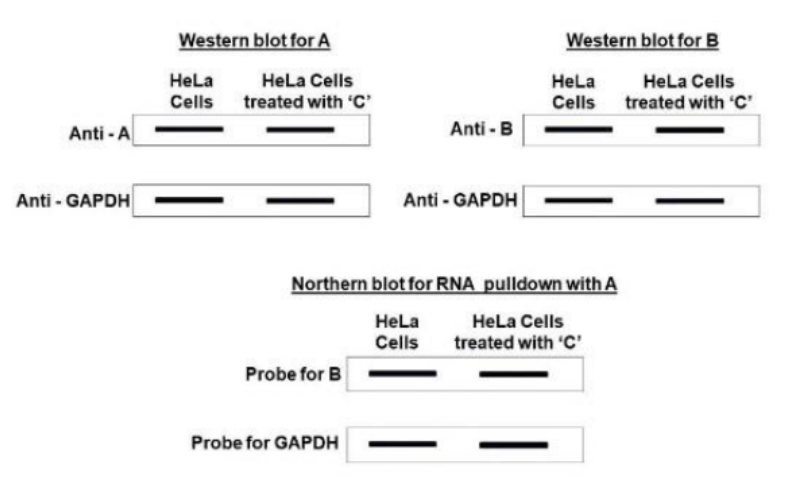
Correct option is C
Immunoprecipitation (IP):
A technique that uses antibodies to capture a specific protein (the target protein) from a sample. This allows for the study of the protein's interactions, modifications, or functions.
Process: The sample (e.g., cell lysate) is incubated with an antibody specific to the target protein, and then a secondary reagent (like Protein A-Sepharose) is used to pull down the antibody-protein complex. The bound proteins are then analyzed, often using techniques like Western blotting.
Importance of Controls: Proper controls are essential in IP experiments to confirm that the observed interactions are due to specific binding and not due to nonspecific interactions or artifacts. Controls help to identify such nonspecific interactions.
Nonspecific Binding: If the beads (e.g., Protein A-Sepharose) bind other proteins nonspecifically, it can lead to misleading results. Controls help to identify such nonspecific interactions.
Explanation:
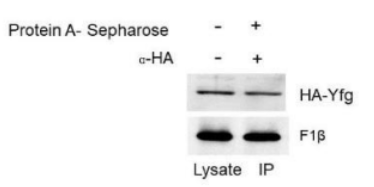
The experiment involves the following components:
HA-Yfg: The protein of interest that is tagged with HA (Hemagglutinin) for detection.
F1β: The protein being tested for interaction with HA-Yfg.
Protein A-Sepharose: A resin used to pull down proteins that are bound to antibodies (in this case, the HA tag).
Control A: Include a lane where α-HA is not added but Protein A-Sepharose is added.
This control checks for any nonspecific binding of the Sepharose beads. If any signal appears in this lane, it indicates that the Protein A-Sepharose is nonspecifically binding proteins from the lysate, thus potentially misleading the results.
Control B: Include a lane where neither α-HA nor Protein A-Sepharose are added.
This serves as a baseline control, indicating the levels of proteins present in the lysate without any interaction or pull-down. However, it does not specifically contribute to confirming the interaction between F1β and HA-Yfg.
Control C: Include a lane where α-HA is added but Protein A-Sepharose is not added.
This checks if F1β can be detected without the pull-down mechanism. It’s useful for determining if the detection method itself is functional, but it does not directly validate the interaction.
Control D: Include a lane where α-Myc is added instead of α-HA before the addition of Protein A-Sepharose.
This control assesses whether the association seen with α-HA is specific to HA-Yfg. If no interaction occurs with α-Myc, it suggests that F1β does not interact with proteins other than those tagged with HA.
Negative Control (Control A):
By including a lane where α-HA is absent but the beads are present, researchers can check for nonspecific binding. If any protein is detected in this lane, it indicates that the signal observed in other lanes might not solely be due to the specific interaction with the target protein.
Specificity Control (Control D):
By using an unrelated antibody (like α-Myc) in place of the target antibody (α-HA), researchers can determine if the target protein (F1β) interacts specifically with the antibody. If no interaction is observed with α-Myc, it supports the conclusion that the binding seen with α-HA is indeed specific to HA-Yfg.