Correct option is B
The Langmuir adsorption model explains adsorption by assuming an adsorbate behaves as an ideal gas at isothermal conditions. According to the model, adsorption and desorption are reversible processes. This model even explains the effect of pressure; i.e., at these conditions the adsorbate's partial pressure
is related to its volume V adsorbed onto a solid adsorbent. The adsorbent, as indicated in the figure, is assumed to be an ideal solid surface composed of a series of distinct sites capable of binding the adsorbate. The adsorbate binding is treated as a chemical reaction between the adsorbate gaseous molecule
and an empty sorption site S. This reaction yields an adsorbed species
with an associated equilibrium constant
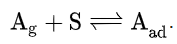
The Langmuir adsorption equation is
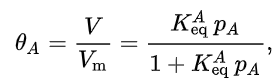
where is the fractional occupancy of the adsorption sites, i.e., the ratio of the volume V of gas adsorbed onto the solid to the volume of a gas molecules monolayer covering the whole surface of the solid and completely occupied by the adsorbate. A continuous monolayer of adsorbate molecules covering a homogeneous flat solid surface is the conceptual basis for this adsorption mode.












