Correct option is A
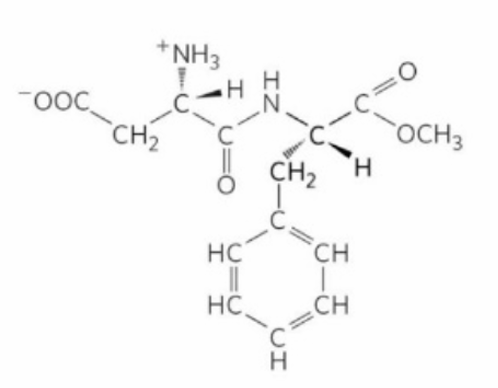
The given structure represents a peptide formed between L-Aspartic acid and L-Phenylalanine. In this structure, both amino acids are in their L configurations. The dipeptide is formed by Peptide bond between the Carboxyl group of L-Aspartic acid and the amino group of L-Phenylalanine. The methyl ester group is attached to the carboxyl group of the phenylalanine residue. This is consistent with option 1.
Information Booster:
L-amino acids are the most commonly found in naturally occurring proteins.
The peptide bond is formed between the amino group of one amino acid and the carboxyl group of another.
L-Aspartic acid has a side chain with a carboxyl group (-COOH), which is positioned as shown in the structure.
L-Phenylalanine has a benzene ring as its side chain, which is depicted correctly in the structure.
In this configuration, the L-form of both amino acids is crucial for correct peptide bonding and three-dimensional structure.
The structure shows the correct spatial orientation and stereochemistry for both L-Aspartyl and L-Phenylalanine.
Additional Knowledge:
Option 2: D-Aspartyl-L-phenylalanine methyl ester – This configuration involves D aspartic acid, which is not shown in the structure.
Option 3: D-Aspartyl-D-phenylalanine methyl ester – This configuration involves both amino acids in the D form, which is inconsistent with the structure shown.
Option 4: L-Aspartyl-D-phenylalanine methyl ester – This configuration involves L aspartic acid and D phenylalanine, which does not match the structure.