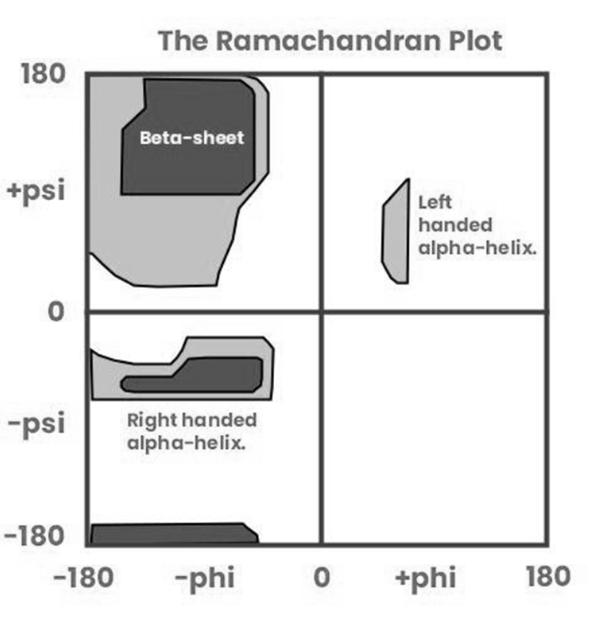
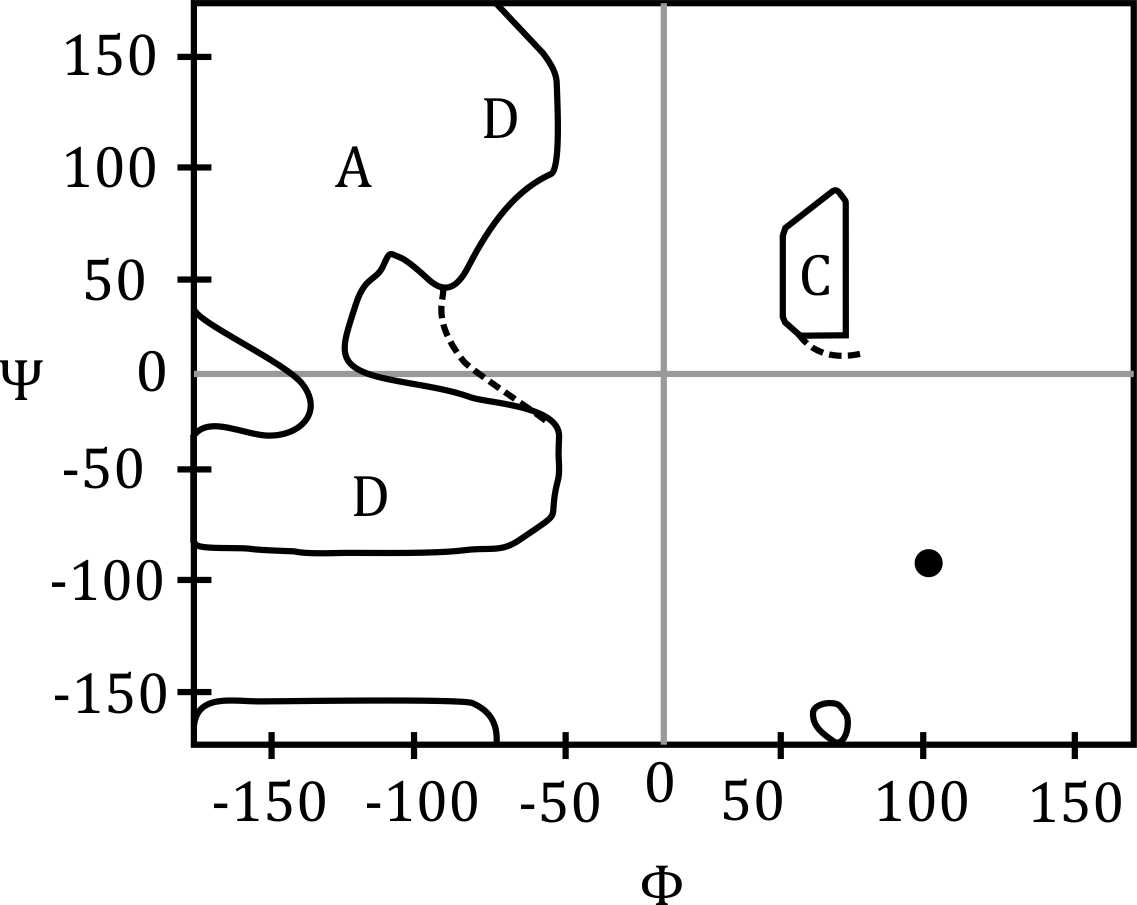
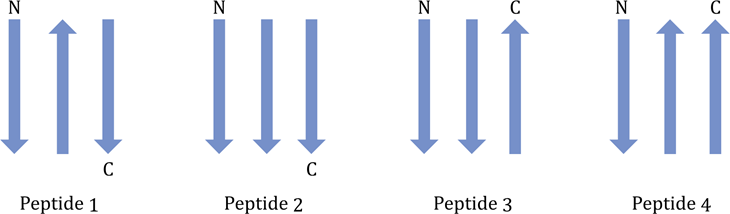
Correct option is A
The correct answer is Mass spectrometry. Mass spectrometry (MS) is the most powerful and accurate technique to identify modifications such as acetylation on a lysine residue of a protein. In this technique, proteins are ionized and analyzed based on their mass-to-charge ratio. The presence of acetylation on the lysine residue causes a change in the mass of the peptide, which can be detected and quantified. MS can identify specific post-translational modifications (PTMs) like acetylation, phosphorylation, or methylation on proteins with high sensitivity and precision.
Information Booster:
- Acetylation is a common post-translational modification (PTM) that typically occurs on lysine residues of proteins. In mass spectrometry, acetylation can be identified because the acetyl group adds a distinct mass (42 Da) to the lysine residue. This modification alters the peptide's mass, allowing it to be distinguished from unmodified peptides during the MS analysis.
- Mass spectrometry can provide detailed information not only on the presence of acetylation but also on the site of modification and the extent of modification in the protein.
Additional Information:
- SDS-PAGE: This technique is used for separating proteins based on their size (molecular weight). While it can help analyze protein expression or purity, it does not detect specific post-translational modifications like acetylation.
- Light scattering: This technique is mainly used to measure the size and shape of molecules in solution, but it does not provide detailed information about specific chemical modifications such as acetylation.
- CD spectroscopy: Circular Dichroism (CD) spectroscopy is used to study the secondary structure of proteins by measuring their chiral properties. While it can provide information on the overall folding of a protein, it does not directly identify specific modifications like acetylation on lysine residues.