Correct option is A
Explanation-
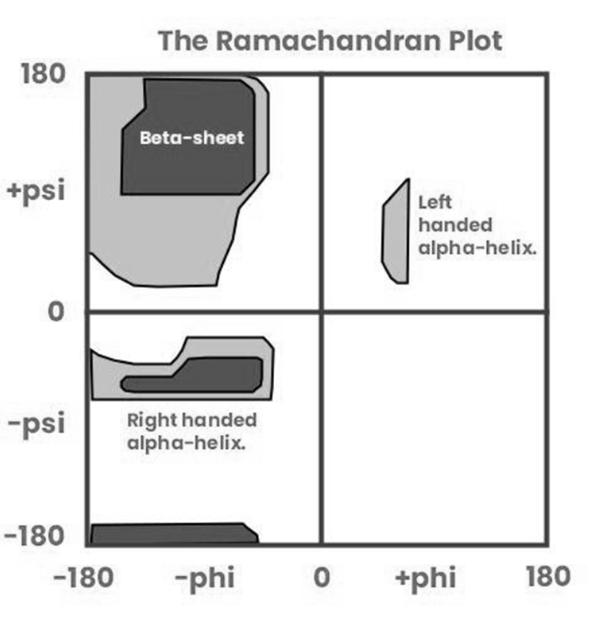
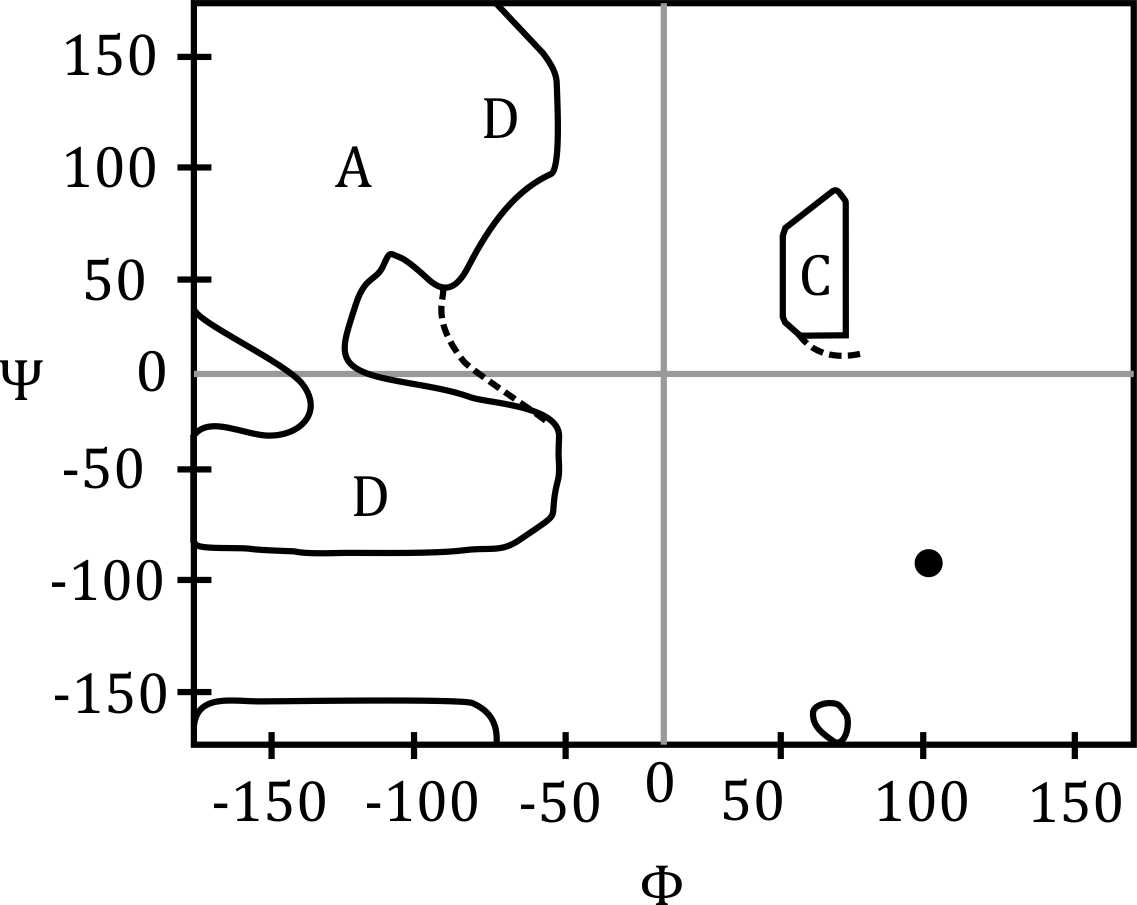
The Ramachandran plot shows which combinations of backbone torsional angles φ and ψ are allowed for an amino acid residue in a protein.
Correct Reason: Steric Hindrance
"Two atoms cannot occupy the same space" — this is a fundamental principle of chemistry and physics, known as the Pauli exclusion principle and steric hindrance.
Each amino acid in a polypeptide has atoms that take up space. When φ and ψ angles are rotated into certain positions, atoms may clash or come too close to each other. Such combinations are energetically unfavorable or physically impossible.
Therefore, only specific combinations of φ and ψ allow enough space between atoms — and these combinations are shown as allowed regions on the Ramachandran plot (like beta-sheets, right-handed alpha helices, etc.)
Incorrect Option -
Option b: The geometry of the peptide bond is planar, but that alone doesn’t explain why all φ and ψ combinations are restricted — steric clashes are the main reason.
Option c: Beta sheets and alpha helices occupy allowed regions, but they don’t define the allowed space — it's the steric constraints that define it.
Option d: Tertiary folding happens after the local φ and ψ angles are set — it doesn’t determine their basic allowed values.
Final Answer:
Option a — "Two atoms cannot occupy the same space."