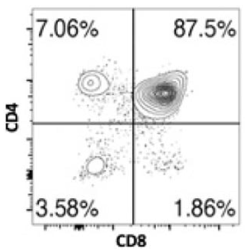
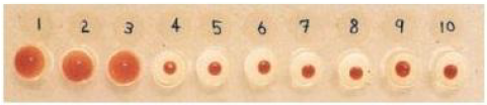
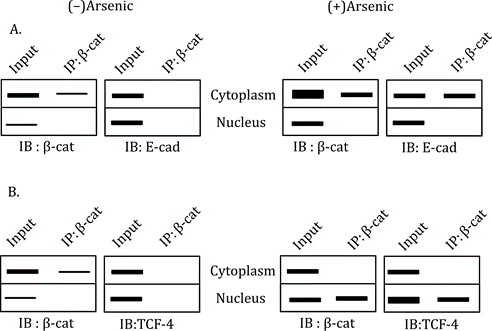
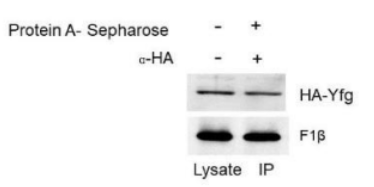
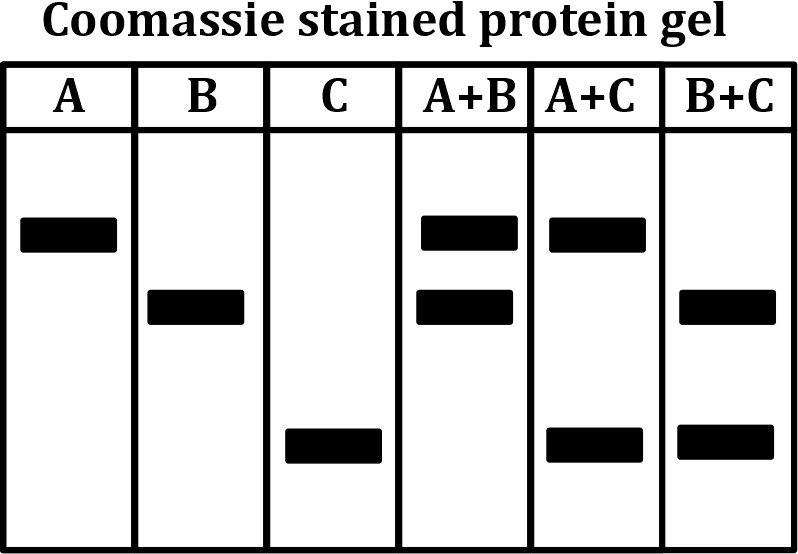
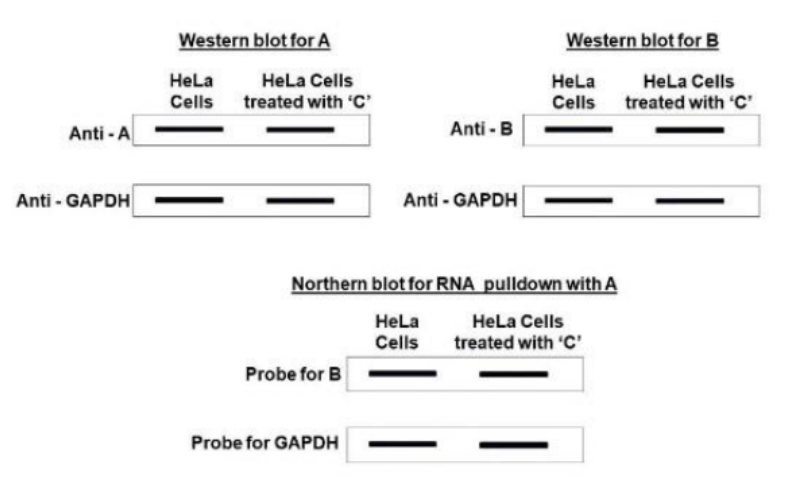
Correct option is C
Option 3: The slope of the standard curve generated by student B was lower than optimal.
- Explanation:
- The slope of the standard curve in an ELISA test is crucial because it determines the sensitivity of the test. If the slope is too flat (lower than optimal), the test will have reduced sensitivity, meaning small changes in concentration may not result in corresponding changes in absorbance.
- A lower slope could cause Student B's measurements to be inaccurately high because their data points would not be as finely resolved, leading to exaggerated values for concentrations. This could explain why Student B reported a much higher concentration (450 μg/ml) than Student A's correct concentration (100 μg/ml).
- Essentially, a less steep slope means that the relationship between absorbance and concentration is weaker, leading to higher reported concentrations, particularly when absorbance values are in the lower range of the standard curve.
Why the other options are less likely:
Option 1: If the plate was not washed properly between coating and blocking, there might be non-specific binding, but this would likely result in high background noise rather than a significant overestimation of the concentration like what was observed for Student B.
Option 2: Improper washing after the addition of the sample would cause false positive results, but this would generally increase background noise, not cause such a significant overestimation of concentration.
Option 4: If the negative control showed very little absorbance, it suggests the assay worked well to detect absence of binding. It would not explain why Student B’s result was higher than expected.